This is an expansion of the Cognitive Atomization concept. In essence, this essay argues that conditioned memory frameworks guide the way we perceive the world, the way we think about the world, our behaviors and our ability to navigate the external world. The NMDAr activity reducing drugs may reduce access to these guiding frameworks and influence them on a spectrum that is dose-dependent. Lower doses may cut our ability to encode new memories, while higher doses may shut down the ability to guide our world models through our historic mental references, in other words, everything that we have learned may be disrupted, causing us to be unfamiliar with the external world and even our own bodies.
ABSTRACT
NMDA receptor antagonists have been researched in memory function due to the way NMDArs are involved in long-term potentiation of neurons. NMDArs have also been explored in research on summation, attention blocking, pain, and schizophrenia. This paper is a research proposal for studying NMDAr antagonists and their effects on memory and perception. The ‘hallucinogenic’ and psychotomimetic properties of NMDAr antagonists may be due to the way perception relies on long-term potentiation (LTP), summation, and coincidence detection in order to form reasonable perceptual models of the external world. A failure of these processes due to impairment of NMDAr binding may result in distinct problems that reveal the role of NMDAr in general cognition, memory, and perception. The proposed reversal of associative patterns and conditioned cognitive responses is termed ‘cognitive atomization’.
Optical illusions are explored as a form of perceptual memory and visual conditioning, as a kind of perceptual auto-correct. Illusions may be associative conditioning in which external sensory cues associate with perceptual representations that are elicited by the cues after learning. The immunity to illusions in schizophrenia is then argued to be similar to visual agnosia, claiming object recognition and perceptual representations are kind of ‘illusions’. This is significant as visual agnosia is also a sign of Alzheimer’s disease. Next, the biology of schizophrenia is compared with Alzheimer’s disease, showing many similarities.
Here I propose a series of tests to determine whether NMDAr antagonists inhibit retrieval of long-term memory (LTM) by using tasks that involve LTM learning, reward-conditioning, optical illusions, and agnosia. Previous research has found that these drugs do not inhibit LTM although this may be dose-dependent so this study aims to explore whether LTM preservation is dose-dependent or a universal property of NMDAr antagonists.
INTRODUCTION
Hebbian and associative learning are thought to involve and possibly require NMDA receptor stimulation (Glanzman 2005). The NMDA receptor (NMDAr) is an ubiquitous glutamate receptor in the brain. This receptor is involved in memory, learning, and perception. Drugs that block NMDAr produce hallucinogenic effects and disrupt the normal processing of the senses (Krystal, D’Souza, Mathalon et al 2003). These drugs are clinically used as anesthetics due to their ability to numb tactile sensation and even elicit dissociative ‘unconscious’ states. NMDAr is thought to be involved in long term potentiation, long term memory, Hebbian learning, coincidence detection and classical conditioning (Tabone & Ramaswami 2012). The brain may utilize this mechanism as a general cellular memory tool. Importantly, NMDAr is found to be a mechanism that is impaired in schizophrenia (Coyle 2012). Here we explore the possibility that blocking NMDA receptors might reverse previously learned associations acutely, so that the world is interpreted non-associatively. Another possibility is that only short-term association is disrupted, where events cannot be detected as coincident. The third possibility is that neither of these are true. If NMDAr blockade reverses previously learned associations, then this would help in understanding how cognitive and perceptual changes manifest from NMDAr antagonists and in schizophrenia. Here I propose a series of tests to unveil whether the effects of higher doses of the NMDAr antagonist known as dextromethorphan (DXM) have on memory functions including short-term memory, long-term memory, perception, illusions and summation. These tests will look at functional magnetic resonance imaging (fMRI) to determine which regions may be altered by the drug, where I predict changes to hippocampal activity as well as cortical activity.
LTP AND COINCIDENCE
NMDAr has been researched as a memory mechanism due to its’ involvement in long-term potentiation (LTP) (Glanzman 2005). LTP is when a neuron becomes highly sensitive to excitatory transmission for days or weeks. LTP occurs after recurring stimulation of glutamate activity that leads to magnesium release, unblocking NMDAr and which eventually leads to LTP`. The function of NMDAr in learning may be due to the way it has a longer activation cycle compared to the other common glutamate subtype known as AMPA receptor (AMPAr) (Luscher & Malenka 2012). The longer firing cycle may create neural environments where neurons are much more likely to be activated simultaneously rather than in an atomized way. This may help create more integrated signaling of neurons and allow activity to be associated through its simultaneity. On the other hand, AMPAr has a far shorter firing cycle, which could mean that synchronized neuron firing is much rarer. NMDAr has been studied as a ‘coincidence detection’ mechanism in flies (Tabone & Ramaswami 2012). It may be that NMDAr blockade disrupts ‘coincidence’ by reducing simultaneity, leading to events being non-associated.
AMPAr is an ion channel that glutamate binds to and opens the flow of ions, allowing for excitation of the neuron. When AMPAr is bound, NMDAr is usually also opened but it is blocked by magnesium ions which prevent depolarization or any influence on the neuron’s excitation. Magnesium can unbind from the NMDAr which allows sodium and calcium to enter and produce excitation further. So recurrent AMPAr stimulation by glutamate allows more time for magnesium to escape the NMDAr which makes NMDAr open for further neuron excitation by influx of calcium or sodium ions. If calcium enters the cell, LTP can occur, making the neuron sensitive to excitatory transmission in the long term (Luscher & Malenka 2012). NMDAr blocking drugs prevent LTP but also slow its’ decay (Villarreal & Haddad 2002). This is significant as LTP and NMDAr are thought to be critical for associative learning. LTP’s role may be in sensitizing future activation of the learned neural patterns.
Individuals with schizophrenia are thought to have impaired LTP (Tang, Yang, Chen 2009) and associative learning (Hall, Romaniuk, McIntosh, et al 2009) both.
SUMMATION
There is another key concept known as summation. This includes both temporal and spatial summation. The idea is that neuron events that occur within a time window can lead to summation response of these events. For example, two neurons may stimulate the same third neuron within a window of time and produce a combined effect on the third neuron. This is spatial summation. Temporal summation is when a neuron is stimulated repeatedly in a short window of time and produces an enhanced excitatory effect (Felten, O’Banion, Maida, & Netter, 2016). Both spatial and temporal summation interplay together here. NMDAr is thought to be play a role in summation (Wu, Ma, Kelly 2004). Since NMDAr produces prolonged excitatory effects on the neuron compared to AMPAr, it widens the temporal window for which summation can occur. This is supported by research on pain summation which has used the NMDAr antagonist dextromethorphan (DXM) to suppress this summation effect (Price, Mao, Frenk, & Mayer, 1994). Summation seems to be similar to coincidence detection on some level, both involving NMDAr and widening of the time window for simultaneous signaling. Cognitive atomization in this context could be described as desummation.
In schizophrenia, there is impairment of something known as prepulse inhibition. This is a test that involves presenting a series of stimuli to a subject where a smaller pulse sound is given before a loud and startling noise. Non-schizophrenic subjects will usually have a suppressed reaction to the second more startling noise, while schizophrenics do not suppress this (Ludewig, Geyer, & Vollenweider, 2003). One study looked at the way summation and prepulse inhibition are connected (Blumenthal, 1995), which makes sense as NMDAr antagonists disrupt both.
CLASSICAL CONDITIONING
NMDAr may be a crucial aspect of classical conditioning, perhaps both for encoding and retrieval, a hypothesis I aim to test here. Dopamine is classically known as a key component to classical conditioning. Recently research has shown that NMDAr co-activation with D1 dopamine receptors may be crucial to classical conditioning (Baldwin, Sadeghian, & Kelley, 2002) and (Touzani, Bodnar, & Sclafani, 2013). Endogenous opioids such as endorphins, enkephalins, and dynorphins are also known to play a role in reward and aversion conditioning. Endorphins and enkephalins seem to mostly enhance reward conditioning by increasing dopamine activity (Devine, Leone Pocock, & Wise, 1993) and enhancing NMDAr activity via mu-opioid receptor (MOR) binding (Trujillo 2002).
On the other hand, dynorphin is involved in aversion (Land, Bruchas, Lemos et al 2008), fear learning (Bilkei-Gorzo, Erk, Schürmann 2012), and has been thought to play a role in psychotic symptoms in schizophrenia (Clark, & Abi-Dargham 2019). Dynorphin has been shown to block NMDAr directly in rats (Chen, Gu, & Huang, 1995) and binds to kappa-opioid receptors (KOR), which has been shown to reduce dopamine acitivity (Tejeda & Bonci, 2019). KOR activity has also been associated to the aversive component of stress (Land, Bruchas, Lemos et al 2008) and pain (Massaly, Copits, Wilson-Poe 2019) and has been associated to depression (Knoll, & Carlezon Jr. 2010), anxiety (Crowley, Bloodgood, & Hardaway 2016), and PTSD (Bailey, Cordell, Sobin, & Neumeister 2013). The KOR agonist known as Salvia Divinorum is an intensely potent dissociative hallucinogen (Maqueda, Valle, Addy et al 2015) which may help explain dynorphin’s connection to schizophrenia.
This interplay of glutamate, dopamine, and the opioid system may help explain the mechanisms of classical conditioning. MOR agonists may reinforce the learning process by acting as a reward and stimulating dopamine and NMDAr activity to support the likelihood of NMDAr-D1 co-activation that is thought to underlie classical conditioning, while the KOR agonist dynorphin may act as a punishment and oppose reward conditioning by directly blocking NMDAr and reducing dopamine activity through KOR. Interestingly, schizophrenia has been shown to involve impaired associative learning, decreased MOR density (Ashok, Myers, Marques et al 2019), impaired reward processing but still have retained aversion processing (Cheng, Tang, Li, et al 2012).
Some researchers have been working on the use of ketamine in addiction, where the researchers proposed that ketamine may treat addiction by attenuating reward-cue responses, such as cue-induced blocking (Freeman, Morgan, Pepper et al 2013) and craving (Dakwar, Levin, Foltin, et al 2014). Blocking is the idea that reinforced significant stimuli of attention can be spotlighted by blocking out irrelevant and unimportant stimuli. With addiction, this means blocking out other stimuli in favor of reward-cues.
Considering the concept of summation, you could imagine that two neurons, A and B, feed into neuron C as an integral part of coincidence detection and associative learning. The concept of blocking could be applied here in that neuron C may take precedence over neuron A or B alone after associative learning has taken place. A or B may usually have other options than simply stimulating C, but after conditioning, A or B may bias to stimulate C. The unnecessary acuity of the experience or decision can be reduced away, ignored from consciousness so that our response to A and B are automated as the combined C neural response. In relation to vision, we may only notice C, while ignoring A or B as individual responses. This may occur in many ways so that our perception is viewed as a summated experience, reduced from a more noise-like experience of reality.
The idea of atomization here would be to reverse this tendency for C to take precedence over A or B as individual signals. To explain this in terms of neurons is challenging because there will be situations in which being aware of A, B and C are all useful, and each perception or behavioral reaction could be dissected to such a highly fine grained degree that it could be very difficult to give a realistic example. Each perception or behavior is likely a massive combination of As, Bs, and Cs, that are sometimes on or off, depending on which neurons feed into them, determined by the environmental context. Since NMDAr antagonism seems to attenuate blocking and summation, it may dissolve the biases that pressure C signals, leaving us with an atomized experience of high acuity and of dissected conscious experience that lacks integrated meaningfulness and associations.
You could imagine that a large amount of our experiences are shaped strongly by awareness of mostly ‘Cs’, while we ignore many details (As and Bs) around us, except in contexts that bring these details to attention. In lower doses of NMDAr antagonists this may result in the experience of increasing perceptual acuity for things usually unnoticed, and eventually with higher doses one may notice the inability to notice a lot of previously learned meaning and an inability to recognize familiar stimuli. The effect may introduce noise and remove definition and meaning.
NMDAr ANTAGONISM AND COGNITION
Blocking NMDAr has amnesiac effects in patients, which includes blacking out entirely (Farahmandfar, Akbarabadi, Bakhtazad, & Zarrindast 2017). NMDAr antagonists have also been shown to prevent associative learning (Pallares, Nadal, Silvestre, & Ferre 1995). Some research suggests that blocking NMDAr impairs memory consolidation but not recall. This has been the case in a study in Nature that used ketamine on human subjects, which used doses of 0.27mg/kg. The study showed that learning of new information is impaired but recall of previously learned information is not (Rowland, Astur, Jung 2005). According to PsychonautWiki, this would be considered a threshold-light dose, meaning that it is in the low range of doses, barely producing discernable subjective effects (PsychonautWiki). The notion of cognitive atomization is that the intense ‘hallucinogenic’ and cognitive effects of ketamine are due to the disruption of the recall of learned associations. It could be the case that low doses would impair learning of new associations much earlier than impairing reinforced previously learned associations. Perhaps non-reinforced neurons are more sensitive to being blocked than reinforced neurons.
PsychonautWiki lists ‘memory suppression’ as a distinct effect from amnesia as a side effect of NMDAr antagonists. In the description of this effect it notes that short-term memory is suppressed much earlier than long-term memory. At very high doses, the Wiki suggests that one may even forget who they are, where they live, or even a failure to remember what humans are (PsychonautWiki). It may be that basic sensory information such as how to remain balanced, or identifying the shapes of objects, or even observing relative spatial distances are more prone to memory suppression than complex declarative memories because these declarative memories involve multiple brain systems and layers of complex sensation and are likely less prone to total shutdown from lower doses of NMDAr blockers.
There are sometimes paradoxical effects of NMDAr antagonists. This is possibly due to the fact that they can increase glutamate release (Lorrain, Baccei, Bristow et al 2003) and decrease GABA activity (Homayoun & Moghaddam 2007), which may both potentiate glutamate activity. NMDAr antagonists have been researched as a tool for preventing NMDAr agonist-induced excitotoxicity. Rather strangely, these drugs were shown to sometimes increase excitotoxicity (Ikonomidou & Turski 2002). The same has been shown with the treatment of seizures with NMDAr antagonists, which are linked to high NMDAr activity. While NMDAr antagonists have been shown to act as anticonvulsants, they sometimes paradoxically increase seizures (Bausch, He, & Dong 2010). Because of these frequent paradoxical effects, we should pay special attention to the doses used in research on NMDAr antagonism. We might even expect low doses to promote NMDAr activity indirectly. This may have relevance for the neurotoxicity of higher doses (Farber, Kim, Dikranian et al 2002) as the onset of the dose and the end tail of the dose may result in hyperactive NMDAr effects, while the peak of the effects may attenuate NMDAr more fully.
There has been at least one study showing enhanced memory after stroke when ketamine was applied (Peters, Villasana, & Schnell 2018). Another study found that ketamine enhanced Pavlovian fear memory and prevented extinction (Radford, Park, Jaiswal et al 2018), but it is very important to note that a recent study found that ketamine’s antidepressant effects require desensitization of one of its’ target receptors, the kappa-opioid receptor (KOR) (Jacobson, Simmons, Wulf et al 2020). This is significant because KOR is already known to be involved in fear learning and KOR mRNA correlates with fear learning, while very low KOR mRNA correlates to fear extinction (Knoll, Muschamp, Sillivan et al 2011). This fits the pattern well and might suggest mechanisms other than NMDAr are involved here.
Another hypothesis to propose is that NMDAr antagonism exclusively atomizes working memory. This could lead to impairments in forming a narrative about the context one is in, also leading to a failure of encoding to long-term memory. This would mean someone could easily lose recognition for familiar things that are usually recognized based on their contexts and conditioned expectations. For example, if you are in your room, then you might have an expectation of what objects are present or likely to be present, but if you lost track of where you were, you might also be less quick or able to recognize the objects in your room. If you cannot recognize the objects, you may further lose touch with the fact that you are in the room, leading to further dissociation from reality. The problem with this hypothesis is that it would still suggest that retrieval is impaired, just perhaps indirectly.
One reason that NMDAr antagonists may only seem to impair short-term memory is that this kind of memory is managed by different regions of the brain that are selectively targeted by lower doses. It may also be due to the fact that newer memories are less well-supported and involve less of the brain. The information may be sensitive for its’ recentness but not well-integrated into a larger library of cues and associations. Older memories may involve more layers of support and involve different mechanisms than short-term memories. They may also be more important and ‘guarded’ somehow. Short-term memories naturally quickly fade with time so they are fragile to loss even without the presence of the drug.
A study in Nature that used higher doses of ketamine (24mg/kg) in sheep found that cortical EEG was entirely shut off temporarily (Nicol & Morton 2020). It is also known that higher doses of ketamine can suppress breathing by disrupting brainstem mechanisms (Green, Roback, Kennedy, & Krauss 2011), suggesting that cortical areas are shut down before the brainstem is. This makes sense because there are endogenous NMDAr antagonists like dynorphin which seem to act as anticonvulsants that release during seizures. The study in Nature on sheep mirrors the effects found after seizure, where cortical regions shut down temporarily in an effect known as cortical spreading depression (Rogawski 2008). This may be the effects of the endogenous anti-glutamatergic mechanisms such as dynorphin suddenly ending the seizure but also producing mass shut down of brain areas. It makes sense that regions of the brain are differentially sensitive to the effects of NMDAr antagonism, otherwise endogenous anti-convulsion might result in death. This would support the hypothesis that different brain regions are dose-dependently sensitive to the effects of NMDAr antagonists. Endogenous anti-seizure mechanisms may perhaps be more selective about where they are active, so we would need to explore this further in future research.
ILLUSIONS
Illusions may be able to be viewed as something opposite of cognitive atomization. Cognitive hyper-integration, predictive perception, and an automated representation of the external world.
There are a few illusions that schizophrenics have been shown to sometimes have immunity to. This might suggest involvement of NMDAr in the development of illusions, perhaps as a form of perceptual learning and associative prediction. There are five illusions I found in the research that schizophrenics have immunity to in the literature. This includes the inverted mask illusion (Dima, Roiser, Dietrich et al 2009), the Chubb illusion (Dakin, Carlin, & Hemsley 2005), the Muller-Lyer illusion (Shoshina, Perevozchikova, Konkina et al 2011), the Ebbinghaus illusion (Silverstein, Keane, Feigenson et al 2013), and lastly the ‘three flash’ illusion (Norton, Ongur, Stromeyer III, & Chen 2008). These illusions may be learned/conditioned visual perception ‘habits’ that have formed in response to repeating and consistent patterns of observations, such as basic dimensionality, laws of shapes, distance, motion, and so on. Since reality never presents us with a contradicting perception, our brain learns to automatically assume certain kinds of visual information to exist in the world that doesn’t match with what kind of signals are actually entering the visual system from the external world. For example, the inverted mask illusion reveals that we will perceive a face with inverted 3D data as a regular forward facing face. This is because we never see inverted faces and thus we can assume that the face is facing towards us, as it always is.
The Ebbinghaus Illusion

This may cause us to disregard external information and assume information to be present as a cognitive tool of efficiency. This may reduce the load that processing vision takes on our conscious processing by automating our response to a small set of cues from the external environment by using a kind of perceptual memory recall rather than analyzing the present environment as if it were unfamiliar. It is a form of stereotyping that helps us simplify processing and ignore much of the external data, relying mostly on memories of how the external data is and only regarding very little external information, such as minimal shading or bifocal spatial data.
Presumably, an illusion like the motion after effect relies on more short term kinds of memory and predictions, in which a repeating motion pattern becomes predictable and assumed to continue even when the source of motion ceases. The perceived continuation of motion seems to make the walls or surrounding objects change shape, move and drift. The initial motion source may condition visual processing and induce a learned motion effect that is very short term and fleeting. On the other hand, an illusion like the inverted face mask may not rely on such short term memories, but instead involve long-term memory for face-like 3D structures and their repeating shapes conditioning one’s expectations for 3D shape in the assessment of present moment face-like stimuli. Those with schizophrenia might have impaired long-term learning due to chronically impaired short-term memory mediated by hypofunction of NMDAr. Using NMDAr antagonists to test people’s responses to different kinds of illusions might reveal distinctions between long-term recall versus immediate learning being blocked. If NMDAr antagonism induces an immunity to long-term kind of illusions, this may reveal at least some alteration to recall of long-term visual memory.
Motion After Effect Illusion
Another illusion known as the three flash illusion also tends to not trick those with schizophrenia. This illusion involves a video of 2 visual blips paired with 3 auditory blips. For most people, this video appears to present 3 three visual blips, possibly due to the audio associating to the visual effect. This can be related to summation in which the sound and the audio might be assumed to be associated, as if the blinking causes the sound. Because we hear the 3 sounds clearly, we assume our visual system missed one of the blinks, as these are far harder to notice and occur in such short time periods. In schizophrenia, this may impaired much like associative learning is impaired. The visual and auditory events may appear to occur as distinct events, non-associated, and essentially atomized into separate experiential pieces. This seems to relate to the concepts of summation and prepulse inhibition, which both involve temporally close events being associated and reacted to and may rely on NMDAr activation. If a loss of NMDAr activity results in such an atomized experience, it could hypothetically impair cue-responses that depend on summated cues. So for example, the recognition of some object may require that lower layers of perception form associations, such as the surfaces that associate to create that 3D object. If one does not recognize lower layers as associated, maybe there is an absence of the cue for recognizing a familiar object. This may open the door to misperceptions. It is worth noting that perhaps objects will often still be recognized but require more processing and involve less perceptual shortcuts, taking up more space for working memory, ultimately reducing awareness capacity through information overload.
Three-Flash Illusion
It is important to note that cannabis has also been linked to illusion immunity (Koethe, Gerth, Neatby et al 2006). Cannabis has also been shown to induce psychotic symptoms very frequently, even in non-schizophrenic subjects (D’Souza, Sewell, & Ranganathan, 2009). The psychoactive effects could be seen as often being a desirable set of psychotic effects, we should be cautious in defining ‘psychotic symptoms’ as inherently bad reactions to the drug, but rather descriptive reactions to the drug. Another study found that an NMDAr antagonist, MK-801, produces a reduction in sensitivity to the Muller-Lyer illusion in capchaun monkeys (Jacobsen, Barros, & Maior, 2017). The sensitivity to this illusion is thought to differ by culture, urban versus rural (Ahluwalia 1978). This suggests that the illusion is learned and is likely a kind of visual long-term memory. This supports the hypothesis that the effects of NMDAr antagonists involve disruption recall of long-term memory, at least perceptually. The application of the drug seems to reverse visual conditioning or visual learning that produces these illusions.
The Inverted-Mask Illusion

The inverted mask illusion (as well as the other illusions) may involve a conditioned response in which a visual cue made up of associations elicit a learned habitual representation in perception. For the inverted mask illusion this involves a habitual representation that is generated in response to common cues of the physical structures of a face, including lighting and shadows. This can be likened to a perceptual auto-correct system. In text-based autocorrect on mobile devices, we observe the wrong word being recalled based on the expectation and prediction of that word belonging within the sentence’s context. These context words bias the expectation of what word is correct by associating the context to a list of words that you might be typing and choosing the most likely word that matches with the most frequent expected word. We observe these auto-correct systems failing us, which is similar to the way our brain pulls up the wrong perception or as we call it, an illusion. The loss of illusions from NMDAr antagonists and schizophrenia might suggest a loss of predictive perception processes, an absence of memory access to retrieve these predicted perceptions.
AGNOSIA

Psychonaut Wiki also notes an effect of pattern recognition suppression that is commonly reported on NMDAr antagonists. This might explain the lack of the three flash illusion in schizophrenic subjects as this may be a suppression of associating audio and video stimuli together. In a sense, this is a kind of ‘atomization’ effect as well. These two stimuli may normally summate due to close temporal proximity of occurrence. If coincidence detection relies on NMDAr, then blocking NMDAr may prevent awareness of coincident occurrences, such as in the three flash illusion. The Wiki notes that this is very similar to the apperceptive agnosia (PsychonautWiki).
Apperceptive agnosia is the failure to recognize visual objects and a failure of visual coherence. In some sense, all recognized objects could be viewed as illusions of a sort. External reality may not be so ‘representational’ in reality, but representations help us navigate the world. In the same way that illusions fail to occur in schizophrenic subjects, you could imagine that all representational models fall apart at the farthest end of this spectrum of phenomena. In the totally atomized state, visual information may become without recognized patterns, instead with an immense perception of distinct pixels. The spectrum of visual changes that occurs between illusions/hyper-recognition of patterns to totally atomized pixels of perceptual qualia might reveal the structure and organization of perceptual patterns themselves. It may reveal which patterns are more significant than others or the order in which layers of illusions are organized into the kinds of representations that we are used to observing. The NMDAr antagonists may dose-dependently strip away these illusions until we reach a state of total agnosia. Cognitive atomization could be defined as agnosification of the mind in some sense. You could imagine that this occurs not only in perception but with cognition generally.
There is an important consideration and nuance that should be clarified here about illusions and visual distortions. We should distinguish agnosiac perceptions from illusions in future research, because at the moment it appears that visual changes may generally be labeled as ‘illusions’ in some of the research that describes the effects of ketamine or similar drugs. One might view the agnosiac state to be full of ‘illusions’ but the very opposite may be true. Cautiously classifying and distinguishing visual alterations will be important in the future of research on perception.
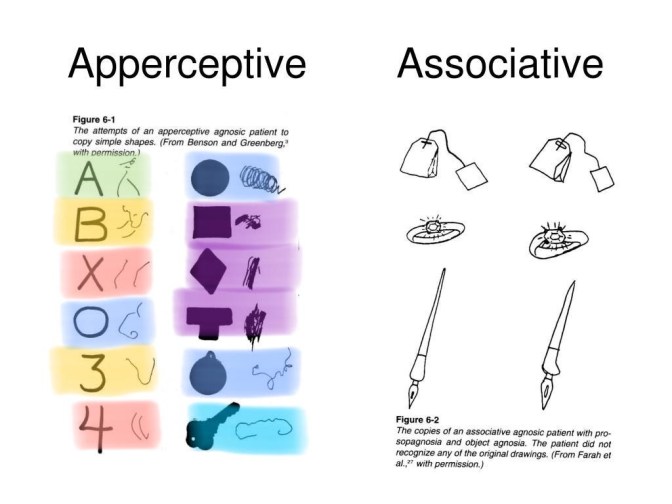
This is an interesting chart that is accessible on Google images. I’ve color labeled objects that appear to have consistencies both in the normal view and the agnosic representation drawing. Notice the X and 4 share the property of being 2 lines. Indeed a normal 4 involves an X shape. The circle representations tend to be strange spirals here. The agnosic A appears to be two lines, much like the 4 and the X, but with a circle representation at the top. 3 and B both normally share the same double curve shape, and the agnosic representations here also share a strange curve-like shape. Perhaps this indicates a failure of spatial summation and the loss of illusions in psychosis could be likened to a very mild form of agnosia. NMDAr antagonists may dose-dependently induce agnosia-like effects.
DEMENTIA
The immunity to illusions may be the first stage in the development of visual agnosia, the first stage in the degradation of object-recognition perception. There is some evidence that the visual processing disorders observed in schizophrenic patients resembles visual agnosia (Gabrovska, Laws, Sinclair, & McKenna 2003). This has particularly interesting implications as visual agnosia is thought to be an indicator in the development of Alzheimer’s disease (Giannakopoulos, Gold, Duc et al 1999).
There are also many overlaps in the biology of both schizophrenia and Alzheimer’s disease (AD). An interesting review has explored some of the overlaps between both schizophrenia and AD, noting that there is a shared atrophy of medial temporal regions, dopaminergic and cholinergic differences, and both share high rates of psychotic symptoms (50% in AD) (White & Cummings 1996). There has been observed a loss of white matter density which was associated to cognitive impairment in both conditions (Kochunov et al 2020).
Tau hyperphosphorylization is thought to be a convergent point between schizophrenia and AD (Deutsch, Rosse & Lakshman 2006). Important to note, AD involves increased beta-amyloid (but it doesn’t seem so with schizophrenia) (Religa, Laudon, Styczynska et al 2003). Both schizophrenia (López-Figueroa, Norton et al 2004) and AD (Lorke, Lu, Cho, & Yew 2006) involve decreased 5HT2a receptor function, which is significant as beta-amyloid injections were found to reduce 5HT2a receptor density (Christensen, Marcussen, Wörtwein et al 2008).
Both schizophrenia and AD (Risser, You, Cairns et al 1996) involve increased dynorphin activity. Dynorphin has been studied as a target for age-related cognitive decline, where the removal of dynorphin genes from animals attenuated age-related cognitive decline (Ménard, Herzog, Schwarzer, & Quirion 2014). This is significant because 5HT2a receptor agonists have been shown to attenuate dynorphin’s depressant effects mediated by KORs (Sakloth, Leggett, Moerke et al 2019). Agonists of nAch alpha7 receptors are thought to be useful in treating both schizophrenia and AD (Toyohara & Hashimoto 2010), which is interesting because this receptor suppresses dynorphin release (Ji et al 2019).
Beta-amyloid also stimulates NMDAr (Texidó, Martín-Satué, Alberdi et al 2011) which has been shown to increase dynorphin release (Hanson, Singh, Merchant et al 1995) and dynorphin’s KOR-mediated depressant effects rely on its’ serotonin reuptake induction through p38 MAPK (Schindler 2013), which suggests a loop may exist here where psychotomimetic and amnesiac effects of dynorphin are prone to increase and serotonin’s impact declines over time. Schizophrenia has been shown to have accelerated brain aging as well (Nenadić, Dietzek, Langbein et al 2017) and used to be known as praecox dementia (Bleuler 1950).
UPDATE: It’s important to note that the research on 5HT2a receptors and schizophrenia is mixed. One meta-analysis from 2006 found no association in post mortem (Li, Duan, & He 2006). Another one from 2014 found a moderate to large effect showing reduced 5HT2a receptor binding both post-mortem studies and with molecular imaging studies, even in unmedicated schizophrenics (Selvaraj, Arnone, Cappai, & Howes 2014). It may be that this association is more complex. It seems highly likely that people with endogenous psychedelic effects would be labeled as schizophrenic along with those experiencing more classic schizophrenic symptoms such as hearing voices. This presents a big problem, but let’s move on.
Perhaps these disorders are not so different, but the narratives and perceptions we have about these disorders guides us to form biased perspectives of each condition. This is not to disregard differences in mechanisms that might exist, after all, the conditions that lead up to each disease state may differ. Further exploration is warranted on this front.
CAPGRAS DELUSION
There is something known as the Capgras delusion, in which people view familiar people as if they were replaced by ‘imposters’. As if some new person inhabits the body of the person. It has been associated with schizophrenia (Silva & Leong 1992), dementia (Kaufman, Newman, & Dawood 2014), and even a temporary case with ketamine (Corlett, D’Souza, & Krystal 2010). Capgras delusion is thought to occur when individuals cannot link together memories and perception, specifically emotional memories (Hirstein & Ramachandran 1997; Merrin & Silberfarb 1976). It may be that ketamine disrupts access to memories, allowing for such delusions to form. This further supports the hypothesis that dissociatives may suppress memory access and adds a beautiful sense of harmony by joining agnosia-like phenomenon, dementia, schizophrenia, and NMDAr antagonists together.
METHODS
There will be five different tests performed in this study. Each test will involve a before-dose and during-dose test to compare the responses to the drug condition. The during-dose test will occur around 1.5 hours after dosing to ensure the drug is active. The drug should last around 4-5 hours for most participants, which will give enough time to perform all the tests.
The first test will explore the participants’ reward conditioned responses and observe if NMDAr antagonism is able to reduce or block conditioned responses. We will condition an aversive response to the participants before the main test takes place. This will occur days prior, to ensure that the conditioned response is definitely a long-term one. Before the application of the drug, their newly developed aversive reflex will be tested to ensure it is still present. The aversive stimuli will be a mild shock that is not physically harmful. This shock will be paired with a noise immediately prior to the shock, by 1 second prior. We expect to observe a conditioned flinch reaction from participants when the sound is elicited, even in the absence of shock. The hypothesis is that NMDAr blockade may attenuate habituated responses. We will also measure heart rate using an iPhone app that tracks heart rate through the camera lens. This can help to detect if heart rate changes in response to the shock and if this change still occurs when the drug is applied. A possible confounder here is that the drug will alter heart rate. This will be measured before and after the shock as well, to see if there is a relative difference.
The second test will involve a similar process but with reward associative learning. The reward conditioning will involve pairing tasty snacks (Oreos and potato chips) with a background soundtrack that has a somewhat unexciting impact on valence. The aim is to associate the near neutral soundtrack with repetitive sessions of delicious snacks. Only one Oreo or chip will be given at a time, with intervals in between so that the participant doesn’t have free access to the snack. We will measure the participants subjective urge/craving intensity on a scale of 1-10. We will play the music without the Oreos or chips, only showing the subject these snacks from a distance and then measure how strongly they urge. The drug condition will follow the same process where music is paired with visual snack stimuli and we will measure the participants’ craving rating.
The third test will be a long-term memory task and involve exposing participants to a short video and asking them to recall a list of details from the film. The participants will be asked misleading questions such as “was there object x?” when the object isn’t present in the film. Free recall of details will be requested as well as recognition of specific details. The video will be shown 4 times on different dates before the drug condition and 1 time during the drug condition test. This will help us test long-term memory retrieval.
In the fourth test we will observe the effects of DXM on the development of the motion after-effect illusion. The participants will watch a 45 second video that is known to induce the motion after effect illusion and each participant will use a provided stop watch that starts as the video stops and the participant allows the timer to go on until their illusion diminishes to baseline again. This will first occur sober and then later after the drug kicks in. Sober controls will also take part in both tests, because the effect might also become weaker the second time. The results will be compared with each other group.
In a fifth test we will observe if the participant’s response to the inverted mask illusion, the Chubb illusion, the Muller-Lyer illusion, the Ebbinghaus illusion, and the ‘three flash’ illusion changes in response to the drug conditions. As a novel test, the motion after-effect illusion will also be tested as a sixth illusion. The hypotheses is that the drug will diminish responsiveness to each illusion in a dose-dependent manner. Each group will be tested before and after the dose takes effect. While it isn’t totally clear how each illusion relates to specific mechanisms, these illusions are often known to be absent in some patients with psychotic disorders. It is possible that these illusions are a product of learning, LTP, or somehow dependent on NMDAr. Since NMDAr is thought to be hypoactive during psychotic states, we will test if NMDAr antagonists block these illusion. If the illusions fail on the NMDAr antagonist dose, then it may be that certain kinds of long-term (and short-term for the motion after-effect illusion) memory is impaired. While we may not traditionally think of perception as involving long-term memories, these illusions may be viewed as a conditioned responses to constant exposure to forward facing faces, while the motion after-effect may represent an immediate development of motion prediction processes.
A visual agnosia test involving images will be given to see if there are any apparent symptoms of agnosia in the subjects. This will be given before the drug condition and also during the drug condition to see if any observable differences emerge. If results suggest agnosic deficits then this will be compared with the prevalence of immunity to illusions to determine if any correlation exists.
An example of an agnosia test.

THE DRUG
The drug DXM will be used in this experiment. There will be four groups, a placebo control group, a low dose (2mg/kg) group, a medium dose (5mg/kg) group, and a high dose (10mg/kg) group. This doses are chosen to reflect William White’s DXM FAQ dosage ‘plateaus’, which White cites each have distinctly different effects (White). These doses represent the first 3 ‘plateaus’. The fourth plateau is ignored due to reducing risks of adverse effects on the subjects.
This drug is chosen because it is legal and available over the counter, making it easier to access. The higher dose should be expected to produce profound alterations to the participants’ conscious state. We will have a medical response team prepared in case of any problematic effects. A low dose of Lorazepam will be handy if the participant wishes to abort. This drug is an anti-anxiety drug that binds to the benzodiazepine sites on GABA neurons, enhancing GABA activity. This may help to end some of the effects of DXM that are caused by impaired GABA activity due to NMDAr blockade.
THE SUBJECTS
The participants will include university students who volunteer and have had previous experience with NMDAr antagonists, such as ketamine, DXM, PCP, or others. This will help to reduce problematic reactions that might occur from naïve users who are shocked by the effects of the drug. The participants will be given a consent form that lists the possible negative reactions. We will aim to have at least 15 participants and hopefully up to 50. The participants will be gathered through advertisements on the university campus. Each participant will be screened for mental health or physical health problems and no one on medication will be permitted to be a participant. Participants must be at least 24 years old, simply to ensure that there is no contraindication with the developing brain.
Each test will involve the use of the computer video conferencing service known as Zoom. This will allow a wider range of participants from multiple universities. Local students can act as a temporary lab assistants in order to assist with the experiment and participants. Zoom will allow instructions to be given to all participants in synchrony. This setup accommodates the need for social distancing during the pandemic as well.
ANALYSIS
Relationships will be explored between sensitivity to illusion, agnosia, and recall of learned associations, long term memory and dose of DXM to find any correlations. There is special curiosity about whether which effects occur in smaller doses versus larger doses. For example, we might expect lower doses abolish illusion sensitivity while higher doses induce agnosia-like effects. We might expect that the after-effect illusion is lost at lower doses than other illusions that may rely on long term memory as opposed to short term memory. If these expectations are met, it would support a dose-dependent model of the effects of NMDAr antagonists.
Lastly, the inclusion of fMRI is optional based on whether access to the technology is available. If this method is included, we will use the fMRI on each of these tests, during the drug condition tests to compare with placebo groups. We predict changes to hippocampal or generally temporal lobe function may be noted on fMRI in the drug condition that doesn’t occur with the placebo group. There may also be changes to occipital lobe, general cortical, and prefrontal cortex function during the drug condition.
FURTHER IMPLICATIONS
This research has implications for a wide range of brain disorders and learning problems, including schizophrenia, dementia, addiction, and associative learning. The outcome of the study will help to determine if NMDAr is involved solely in consolidation or if it is also involved in retrieval at different doses. Disorders involving disruptions in retrieval may involve NMDAr dysfunction. If NMDAr is critical in long-term memory retrieval, this could have wide-ranging implications for many systems of neurotransmitters that alter NMDAr binding circumstantially, for example D2 receptors (Kotecha, Oak, Jackson et al 2002), D4 receptors (Wang, Zhong, Gu, & Yan 2003), 5HT2a receptors (Dantsuji, Nakamura, Nakayama et al 2019), 5HT2c receptors (Maura, Marcoli, Pepicelli et al 2000), and more. The effects produced by ligands of these receptor types may be partially due to their impact on NMDAr function, either enhancing learning processes by accelerating them or reducing learning or reducing access to learned/conditioned neural responses. If NMDAr has no role in the retrieval of learned/conditioned responses, then it may support the notion that NMDAr is only involved in consolidation.
If we discover that NMDAr is key to associative brain processing, it means that we could observe the organization of how learned associations and learned neural patterns are structured based on the dose-dependent reversal of some associations and not others. It is important to figure out what dose-dependency of this effect is representing. For example, are NMDAr antagonists revealing the layers of these cognitive constructs based on how they are organized? Is the brain organized in layers that are impacted in order of their structure? Each region might be expected to have a different dose-dependent responsiveness as well.
We may be able to view the balance dis-equilibrium effects of NMDAr blockade as an effect of removing learned sense of gravity. Meaning that we use memory mechanisms to keep our balance. This makes sense because balance likely must involve prediction and expectations for tuning motor movements to compensate for sudden changes. Similarly, the failure of eye tracking that occurs on NMDAr antagonists may be due to inhibiting the conditioned ability to track objects. Infants are not born with the ability to track objects, instead this seems to be a learned skill.
The results of the three flash illusion may give more direct evidence of problems observing events as associated. The test seems to be simplistically a product of temporally associated stimuli and may imply involvement of temporal summation. This may mean that coherent non-agnosic perception requires summation. This is also likely related to coincidence detection, which may utilize summation. The blockade of summation by NMDAr antagonists may also be a blockade of coincidence detection.
. . .
Special thanks to the six patrons: Melissa Bradley, Morgan Catha, Niklas Kokkola, Abhishaike Mahajan, Riley Fitzpatrick, and Charles Wright! Abhi is also the artist who created the cover image for Most Relevant. Please support him on instagram, he is an amazing artist! I’d also like to thank Annie Vu, Chris Byrd, and Kettner Griswold for your kindness and making these projects and the podcast possible through your donations.
If you liked this, follow me on
If you’d like to support these projects check out this page.

CITATIONS
- Ahluwalia, A. (1978). An intra‐cultural investigation of susceptibility to ‘perspective’ and ‘non‐perspective’spatial illusions. British Journal of Psychology, 69(2), 233-241.
- Alkire, M. T., Hudetz, A. G., & Tononi, G. (2008). Consciousness and Anesthesia. Science, 322(5903), 876-880. doi:10.1126/science.1149213 UNUSED CITATION
- Ashok, A. H., Myers, J., Marques, T. R., Rabiner, E. A., & Howes, O. D. (2019). Reduced mu-opioid receptor availability in schizophrenia revealed with [11 C]-carfentanil positron emission tomographic Imaging. Nature communications, 10(1), 1-9.
- Bailey, C. R., Cordell, E., Sobin, S. M., & Neumeister, A. (2013). Recent progress in understanding the pathophysiology of post-traumatic stress disorder. CNS drugs, 27(3), 221-232.
- Baldwin, A. E., Sadeghian, K., & Kelley, A. E. (2002). Appetitive instrumental learning requires coincident activation of NMDA and dopamine D1 receptors within the medial prefrontal cortex. Journal of Neuroscience, 22(3), 1063-1071.
- Bast, T. (2005). Distinct Contributions of Hippocampal NMDA and AMPA Receptors to Encoding and Retrieval of One-Trial Place Memory. Journal of Neuroscience, 25(25), 5845-5856. doi:10.1523/jneurosci.0698-05.2005 UNUSED CITATION
- Bausch, S. B., He, S., & Dong, Y. (2010). Inverse relationship between seizure expression and extrasynaptic NMDAR function following chronic NMDAR inhibition. Epilepsia, 51, 102-105.
- Bilkei-Gorzo, A., Erk, S., Schürmann, B., Mauer, D., Michel, K., Boecker, H., … & Zimmer, A. (2012). Dynorphins regulate fear memory: from mice to men. Journal of Neuroscience, 32(27), 9335-9343.
- Bleuler, E. (1950). Dementia praecox or the group of schizophrenias.
- Blumenthal, T. D. (1995). Prepulse inhibition of the startle eyeblink as an indicator of temporal summation. Perception & Psychophysics, 57(4), 487-494.
- Chen, L., Gu, Y., & Huang, L. Y. (1995). The opioid peptide dynorphin directly blocks NMDA receptor channels in the rat. The Journal of physiology, 482(3), 575-581.
- Cheng, G. L., Tang, J. C., Li, F. W., Lau, E. Y., & Lee, T. M. (2012). Schizophrenia and risk-taking: impaired reward but preserved punishment processing. Schizophrenia research, 136(1-3), 122-127.
- Christensen, R., Marcussen, A. B., Wörtwein, G., Knudsen, G. M., & Aznar, S. (2008). Aβ (1–42) injection causes memory impairment, lowered cortical and serum BDNF levels, and decreased hippocampal 5-HT2A levels. Experimental Neurology, 210(1), 164-171.
- Clark, S. D., & Abi-Dargham, A. (2019). The role of dynorphin and the kappa opioid receptor in the symptomatology of schizophrenia: A review of the evidence. Biological psychiatry, 86(7), 502-511.
- Corlett, P. R., D’Souza, D. C., & Krystal, J. H. (2010). Capgras syndrome induced by ketamine in a healthy subject. Biological psychiatry, 68(1), e1-e2.
- Coyle, J. T. (2012). NMDA Receptor and Schizophrenia: A Brief History. Schizophrenia Bulletin, 38(5), 920-926. doi:10.1093/schbul/sbs076
- Crowley, N. A., Bloodgood, D. W., Hardaway, J. A., Kendra, A. M., McCall, J. G., Al-Hasani, R., … & Lowell, B. B. (2016). Dynorphin controls the gain of an amygdalar anxiety circuit. Cell reports, 14(12), 2774-2783.
- D’Souza, D. C., Sewell, R. A., & Ranganathan, M. (2009). Cannabis and psychosis/schizophrenia: human studies. European archives of psychiatry and clinical neuroscience, 259(7), 413-431.
- Dakin, S., Carlin, P., & Hemsley, D. (2005). Weak suppression of visual context in chronic schizophrenia. Current Biology, 15(20), R822-R824.
- Dakwar, E., Levin, F., Foltin, R. W., Nunes, E. V., & Hart, C. L. (2014). The Effects of Subanesthetic Ketamine Infusions on Motivation to Quit and Cue-Induced Craving in Cocaine-Dependent Research Volunteers. Biological Psychiatry, 76(1), 40-46. doi:10.1016/j.biopsych.2013.08.009
- Dantsuji, M., Nakamura, S., Nakayama, K., Mochizuki, A., Park, S. K., Bae, Y. C., … & Inoue, T. (2019). 5‐HT2A receptor activation enhances NMDA receptor‐mediated glutamate responses through Src kinase in the dendrites of rat jaw‐closing motoneurons. The Journal of physiology, 597(9), 2565-2589.
- Devine, D. P., Leone, P. A. O. L. A., Pocock, D. O. R. O. T. H. Y., & Wise, R. A. (1993). Differential involvement of ventral tegmental mu, delta and kappa opioid receptors in modulation of basal mesolimbic dopamine release: in vivo microdialysis studies. Journal of Pharmacology and Experimental Therapeutics, 266(3), 1236-1246.
- Deutsch, S. I., Rosse, R. B., & Lakshman, R. M. (2006). Dysregulation of tau phosphorylation is a hypothesized point of convergence in the pathogenesis of alzheimer’s disease, frontotemporal dementia and schizophrenia with therapeutic implications. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 30(8), 1369-1380.
- Dima, D., Roiser, J. P., Dietrich, D. E., Bonnemann, C., Lanfermann, H., Emrich, H. M., & Dillo, W. (2009). Understanding why patients with schizophrenia do not perceive the hollow-mask illusion using dynamic causal modelling. Neuroimage, 46(4), 1180-1186.
- Dissociatives. (2020, January 29). Retrieved June 27, 2020, from https://psychonautwiki.org/wiki/Dissociative
- Farahmandfar, M., Akbarabadi, A., Bakhtazad, A., & Zarrindast, M. R. (2017). Recovery from ketamine-induced amnesia by blockade of GABA-A receptor in the medial prefrontal cortex of mice. Neuroscience, 344, 48-55.
- Farber, N. B., Kim, S. H., Dikranian, K., Jiang, X. P., & Heinkel, C. (2002). Receptor mechanisms and circuitry underlying NMDA antagonist neurotoxicity. Molecular psychiatry, 7(1), 32-43.
- Felten, D. L., O’Banion, M. K., Maida, M. S., & Netter, F. H. (2016). Neurons and Their Properties. In Netter’s Atlas of Neuroscience (pp. 1-42). Philadelphia, PA: Elsevier.
- Freeman, T. P., Morgan, C. J., Pepper, F., Howes, O. D., Stone, J. M., & Curran, H. V. (2013). Associative blocking to reward-predicting cues is attenuated in ketamine users but can be modulated by images associated with drug use. Psychopharmacology, 225(1), 41-50.
- Gabrovska, V. S., Laws, K. R., Sinclair, J., & McKenna, P. J. (2003). Visual object processing in schizophrenia: evidence for an associative agnosic deficit. Schizophrenia research, 59(2-3), 277-286.
- Giannakopoulos, P., Gold, G., Duc, M., Michel, J. P., Hof, P. R., & Bouras, C. (1999). Neuroanatomic correlates of visual agnosia in Alzheimer’s disease: a clinicopathologic study. Neurology, 52(1), 71-71.
- Glanzman, D. L. (2005). Associative Learning: Hebbian Flies. Current Biology, 15(11). doi:10.1016/j.cub.2005.05.028
- Green, S. M., Roback, M. G., Kennedy, R. M., & Krauss, B. (2011). Clinical Practice Guideline for Emergency Department Ketamine Dissociative Sedation: 2011 Update. Annals of Emergency Medicine, 57(5), 449-461. doi:10.1016/j.annemergmed.2010.11.030
- Hall, J., Romaniuk, L., McIntosh, A. M., Steele, J. D., Johnstone, E. C., & Lawrie, S. M. (2009). Associative learning and the genetics of schizophrenia. Trends in neurosciences, 32(6), 359-365.
- Hanson, G. R., Singh, N., Merchant, K., Johnson, M., & Gibb, J. W. (1995). The role of NMDA receptor systems in neuropeptide responses to stimulants of abuse. Drug and alcohol dependence, 37(2), 107-110.
- Hirstein, W., & Ramachandran, V. S. (1997). Capgras syndrome: a novel probe for understanding the neural representation of the identity and familiarity of persons. Proceedings of the Royal Society of London. Series B: Biological Sciences, 264(1380), 437-444.
- Homayoun, H., & Moghaddam, B. (2007). NMDA receptor hypofunction produces opposite effects on prefrontal cortex interneurons and pyramidal neurons. Journal of Neuroscience, 27(43), 11496-11500.
- Ikonomidou, C., & Turski, L. (2002). Why did NMDA receptor antagonists fail clinical trials for stroke and traumatic brain injury?. The Lancet Neurology, 1(6), 383-386.
- Jacobsen, M. E., Barros, M., & Maior, R. S. (2017). MK-801 reduces sensitivity to Müller-Lyer’s illusion in capuchin monkeys. Behavioural Brain Research, 316, 54-58.
- Jacobson, M. L., Simmons, S. C., Wulf, H. A., Cheng, H., Feng, Y. H., Nugent, F. S., … & Lucki, I. (2020). Protracted Effects of Ketamine Require Immediate Kappa Opioid Receptor Activation and Long‐Lasting Desensitization. The FASEB Journal, 34(S1), 1-1.
- Ji, L., Chen, Y., Wei, H., Feng, H., Chang, R., Yu, D., … & Zhang, M. (2019). Activation of alpha7 acetylcholine receptors reduces neuropathic pain by decreasing dynorphin A release from microglia. Brain research, 1715, 57-65.
- Kaufman, K. R., Newman, N. B., & Dawood, A. (2014). Capgras delusion with violent behavior in Alzheimer dementia: Case analysis with literature review. , 26, 2, 26(2), e9-e13.
- Ketamine. (2020, June 05). Retrieved June 27, 2020, from https://psychonautwiki.org/wiki/ketamine
- Knoll, A. T., & Carlezon Jr, W. A. (2010). Dynorphin, stress, and depression. Brain research, 1314, 56-73.
- Knoll, A. T., Muschamp, J. W., Sillivan, S. E., Ferguson, D., Dietz, D. M., Meloni, E. G., … & Carlezon Jr, W. A. (2011). Kappa opioid receptor signaling in the basolateral amygdala regulates conditioned fear and anxiety in rats. Biological psychiatry, 70(5), 425-433.
- Kochunov, P., Zavaliangos-Petropulu, A., Jahanshad, N., Thompson, P. M., Ryan, M. C., Chiappelli, J., … & Sampath, H. (2020). A White Matter Connection of Schizophrenia and Alzheimer’s Disease. Schizophrenia Bulletin.
- Koethe, D., Gerth, C. W., Neatby, M. A., Haensel, A., Thies, M., Schneider, U., … & Leweke, F. M. (2006). Disturbances of visual information processing in early states of psychosis and experimental delta-9-tetrahydrocannabinol altered states of consciousness. Schizophrenia research, 88(1-3), 142-150.
- Kotecha, S. A., Oak, J. N., Jackson, M. F., Perez, Y., Orser, B. A., Van Tol, H. H., & MacDonald, J. F. (2002). A D2 class dopamine receptor transactivates a receptor tyrosine kinase to inhibit NMDA receptor transmission. Neuron, 35(6), 1111-1122.
- Krystal, J. H., D’Souza, D. C., Mathalon, D., Perry, E., Belger, A., & Hoffman, R. (2003). NMDA receptor antagonist effects, cortical glutamatergic function, and schizophrenia: toward a paradigm shift in medication development. Psychopharmacology, 169(3-4), 215-233.
- Land, B. B., Bruchas, M. R., Lemos, J. C., Xu, M., Melief, E. J., & Chavkin, C. (2008). The dysphoric component of stress is encoded by activation of the dynorphin κ-opioid system. Journal of Neuroscience, 28(2), 407-414.
- Li, D., Duan, Y., & He, L. (2006). Association study of serotonin 2A receptor (5-HT2A) gene with schizophrenia and suicidal behavior using systematic meta-analysis. Biochemical and biophysical research communications, 340(3), 1006-1015.
- López-Figueroa, A. L., Norton, C. S., López-Figueroa, M. O., Armellini-Dodel, D., Burke, S., Akil, H., … & Watson, S. J. (2004). Serotonin 5-HT1A, 5-HT1B, and 5-HT2A receptor mRNA expression in subjects with major depression, bipolar disorder, and schizophrenia. Biological psychiatry, 55(3), 225-233.
- Lorke, D. E., Lu, G., Cho, E., & Yew, D. T. (2006). Serotonin 5-HT 2A and 5-HT 6 receptors in the prefrontal cortex of Alzheimer and normal aging patients. BMC neuroscience, 7(1), 36.
- Lorrain, D., Baccei, C. S., Bristow, L. J., Anderson, J. J., & Varney, M. A. (2003). Effects of ketamine and N-methyl-D-aspartate on glutamate and dopamine release in the rat prefrontal cortex: modulation by a group II selective metabotropic glutamate receptor agonist LY379268. Neuroscience, 117(3), 697-706.
- Ludewig, K., Geyer, M. A., & Vollenweider, F. X. (2003). Deficits in prepulse inhibition and habituation in never-medicated, first-episode schizophrenia. Biological psychiatry, 54(2), 121-128.
- Luscher, C., & Malenka, R. C. (2012). NMDA Receptor-Dependent Long-Term Potentiation and Long-Term Depression (LTP/LTD). Cold Spring Harbor Perspectives in Biology, 4(6). doi:10.1101/cshperspect.a005710
- Maqueda, A. E., Valle, M., Addy, P. H., Antonijoan, R. M., Puntes, M., Coimbra, J., … & Barker, S. (2015). Salvinorin-A induces intense dissociative effects, blocking external sensory perception and modulating interoception and sense of body ownership in humans. International Journal of Neuropsychopharmacology, 18(12), pyv065.
- Massaly, N., Copits, B. A., Wilson-Poe, A. R., Hipólito, L., Markovic, T., Yoon, H. J., … & Klaas, A. (2019). Pain-induced negative affect is mediated via recruitment of the nucleus accumbens kappa opioid system. Neuron, 102(3), 564-573.
- Maura, G., Marcoli, M., Pepicelli, O., Rosu, C., Viola, C., & Raiteri, M. (2000). Serotonin inhibition of the NMDA receptor/nitric oxide/cyclic GMP pathway in human neocortex slices: involvement of 5‐HT2C and 5‐HT1A receptors. British journal of pharmacology, 130(8), 1853-1858.
- Memory suppression. (2020, April 04). Retrieved June 27, 2020, from https://psychonautwiki.org/wiki/Memory_suppression
- Ménard, C., Herzog, H., Schwarzer, C., & Quirion, R. (2014). Possible role of dynorphins in Alzheimer’s disease and age-related cognitive deficits. Neurodegenerative Diseases, 13(2-3), 82-85.
- Merrin, E. L., & Silberfarb, P. M. (1976). The Capgras phenomenon. Archives of General Psychiatry, 33(8), 965-968.
- Nenadić, I., Dietzek, M., Langbein, K., Sauer, H., & Gaser, C. (2017). BrainAGE score indicates accelerated brain aging in schizophrenia, but not bipolar disorder. Psychiatry Research: Neuroimaging, 266, 86-89.
- Nicol, A. U., & Morton, A. J. (2020). Characteristic patterns of EEG oscillations in sheep (Ovis aries) induced by ketamine may explain the psychotropic effects seen in humans. Scientific Reports, 10(1). doi:10.1038/s41598-020-66023-8
- Norton, D., Ongur, D., Stromeyer III, C., & Chen, Y. (2008). Altered ‘three-flash’illusion in response to two light pulses in schizophrenia. Schizophrenia research, 103(1-3), 275-282.
- Pallares, M. A., Nadal, R. A., Silvestre, J. S., & Ferre, N. S. (1995). Effects of ketamine, a noncompetitive NMDA antagonist, on the acquisition of the lever-press response in rats. Physiology & behavior, 57(2), 389-392.
- Peters, A. J., Villasana, L. E., & Schnell, E. (2018). Ketamine alters hippocampal cell proliferation and improves learning in mice after traumatic brain injury. Anesthesiology: The Journal of the American Society of Anesthesiologists, 129(2), 278-295.
- Price, D. D., Mao, J., Frenk, H., & Mayer, D. J. (1994). The N-methyl-D-aspartate receptor antagonist dextromethorphan selectively reduces temporal summation of second pain in man. Pain, 59(2), 165-174.
- Radford, K. D., Park, T. Y., Jaiswal, S., Pan, H., Knutsen, A., Zhang, M., … & Choi, K. H. (2018). Enhanced fear memories and brain glucose metabolism (18 F-FDG-PET) following sub-anesthetic intravenous ketamine infusion in Sprague-Dawley rats. Translational Psychiatry, 8(1), 1-12.
- Religa, D., Laudon, H., Styczynska, M., Winblad, B., Näslund, J., & Haroutunian, V. (2003). Amyloid β pathology in Alzheimer’s disease and schizophrenia. American Journal of Psychiatry, 160(5), 867-872.
- Risser, D., You, Z. B., Cairns, N., Herrera-Marschitz, M., Seidl, R., Schneider, C., … & Lubec, G. (1996). Endogenous opioids in frontal cortex of patients with Down syndrome. Neuroscience letters, 203(2), 111-114.
- Rogawski, M. A. (2008). Common pathophysiologic mechanisms in migraine and epilepsy. Archives of neurology, 65(6), 709-714.
- Rowland, L. M., Astur, R. S., Jung, R. E., Bustillo, J. R., Lauriello, J., & Yeo, R. A. (2005). Selective Cognitive Impairments Associated with NMDA Receptor Blockade in Humans. Neuropsychopharmacology, 30(3), 633-639. doi:10.1038/sj.npp.1300642
- Sakloth, F., Leggett, E., Moerke, M. J., Townsend, E. A., Banks, M. L., & Negus, S. S. (2019). Effects of acute and repeated treatment with serotonin 5-HT2A receptor agonist hallucinogens on intracranial self-stimulation in rats. Experimental and clinical psychopharmacology, 27(3), 215.
- Schindler, A. G. (2013). Mechanisms Underlying the Adverse Consequences of Stress: A Role for the Dynorphin/Kappa Opioid Receptor System, p38α MAPK, and the Serotonin Transporter (Doctoral dissertation).
- Selvaraj, S., Arnone, D., Cappai, A., & Howes, O. (2014). Alterations in the serotonin system in schizophrenia: a systematic review and meta-analysis of postmortem and molecular imaging studies. Neuroscience & Biobehavioral Reviews, 45, 233-245.
- Shoshina, I. I., Perevozchikova, I. N., Konkina, S. A., Pronin, S. V., Shelepin, I., & Bendera, A. P. (2011). Features of perception of length of segments under conditions of Ponzo and Müller-Lyer illusions in schizophrenia. IP Pavlova, 61(6), 697-705.
- Silva, A., & Leong, G. B. (1992). The Capgras syndrome in paranoid schizophrenia. Psychopathology, 25(3), 147-153.
- Silverstein, S., Keane, B., Feigenson, K., Wang, Y., Mikkilineni, D., & Paterno, D. (2013). Reduced sensitivity to the Ebbinghaus illusion is state related in schizophrenia. Journal of Vision, 13(9), 1261-1261.
- Tabone, C., & Ramaswami, M. (2012). Is NMDA Receptor-Coincidence Detection Required for Learning and Memory? Neuron, 74(5), 767-769. doi:10.1016/j.neuron.2012.05.008
- Tang, T. T. T., Yang, F., Chen, B. S., Lu, Y., Ji, Y., Roche, K. W., & Lu, B. (2009). Dysbindin regulates hippocampal LTP by controlling NMDA receptor surface expression. Proceedings of the National Academy of Sciences, 106(50), 21395-21400.
- Tejeda, H. A., & Bonci, A. (2019). Dynorphin/kappa-opioid receptor control of dopamine dynamics: Implications for negative affective states and psychiatric disorders. Brain research, 1713, 91-101.
- Texidó, L., Martín-Satué, M., Alberdi, E., Solsona, C., & Matute, C. (2011). Amyloid β peptide oligomers directly activate NMDA receptors. Cell calcium, 49(3), 184-190.
- Touzani, K., Bodnar, R. J., & Sclafani, A. (2013). Glucose-conditioned flavor preference learning requires co-activation of NMDA and dopamine D1-like receptors within the amygdala. Neurobiology of learning and memory, 106, 95-101.
- Toyohara, J., & Hashimoto, K. (2010). α7 Nicotinic receptor agonists: potential therapeutic drugs for treatment of cognitive impairments in schizophrenia and Alzheimer’s disease. The open medicinal chemistry journal, 4, 37.
- Trujillo, K. A. (2002). The neurobiology of opiate tolerance, dependence and sensitization: mechanisms of NMDA receptor-dependent synaptic plasticity. Neurotoxicity research, 4(4), 373-391.
- Villarreal, D. M., Do, V., Haddad, E., & Derrick, B. E. (2002). NMDA receptor antagonists sustain LTP and spatial memory: active processes mediate LTP decay. Nature neuroscience, 5(1), 48-52.
- Wang, X., Zhong, P., Gu, Z., & Yan, Z. (2003). Regulation of NMDA receptors by dopamine D4 signaling in prefrontal cortex. Journal of Neuroscience, 23(30), 9852-9861.
- White, K. E., & Cummings, J. L. (1996). Schizophrenia and Alzheimer’s disease: clinical and pathophysiologic analogies. Comprehensive psychiatry, 37(3), 188-195.
- White, W. E. (2001). THE DEXTROMETHORPHAN FAQ. Retrieved June 27, 2020, from https://www.erowid.org/chemicals/dxm/faq/
- Wu, S. H., Ma, C. L., & Kelly, J. B. (2004). Contribution of AMPA, NMDA, and GABAA receptors to temporal pattern of postsynaptic responses in the inferior colliculus of the rat. Journal of Neuroscience, 24(19), 4625-4634.