The kappa-opioid receptor (KOR) system hasn’t had much attention in the pop psych spotlight yet, but nonetheless, it is a very important part of the electro-neurochemical sack we call the brain. The KOR system seems to be as important as other popular neurotransmitters like dopamine, serotonin, or even glutamate. Here we explore a novel class of hallucinogens and how they differ from other types of hallucinogens.
My friend and psychedelic artist (who did the cover art of this post) Utheraptor has coined the term dysdelia to refer to this less popular class of hallucinogenic drugs, the KOR agonists. I found this term to be greatly appropriate. The term dysdelia comes from the roots dys-, which is defined as ‘bad’, and also -delia, which means ‘to reveal’. Taken together, dysdelia means to reveal the negative, or as I’d like to describe it, darkness revealing. In contrast, the term psychedelic means soul revealing. These are the psychedelics of darkness.
Why should the KOR agonists be referred to as dysdelics?
The KOR system is involved in many very upsetting functions, such as depression, stress, anxiety, pain, suffering, dysphoria, PTSD, anhedonia, and psychosis (Knoll & Carlezon Jr 2010; Crowley et al 2016; Massaly et al 2019; Land et al 2008; Rabellino et al 2018; Clark & Abi-Dargham 2019). The brain’s KOR agonists are known as the dynorphins, kind of like endorphins, except bad instead of good feeling. Rather than playing a role in reward learning, dynorphins play a role in aversion and fear behaviors and memory (Bilkei-Gorzo et al 2012). Dynorphins are sometimes referred to as antirewards (Gold et al 2018). It seems like dynorphins could mediate the aversive affect of very many or even all sufferable experiences, since both pain and stress aversive affect seem to be mediated by dynorphins (Massaly et al 2019; Land et al 2008).
Dynorphins are like the demons in our body. After ketamine was observed to require desensitization of the KOR system in order for its’ antidepressant effects to take hold, I decided that ketamine’s effects are a chemical exorcism. This term may even better apply to dysdelics, although there isn’t enough research on them yet.
The official drug of 2020.

The most popularly known dysdelic is Salvia Divinorum. From the salvia-inspired art above, you see a depiction of the body unzipping, ripping apart, then being diced into small pieces, while the head is squashed and spread out into pasta. If it isn’t clear already, most people don’t actually enjoy salvia all that much. But for some, this drug offers an entirely novel mindscape of dysdelia that seems to be a previously unexplored front of the chemical mind.
While there are other dysdelic drugs, such as pentazocine and ketazocine, they have not been used very much, or at least there are not many user reports on the internet. Dysdelics are reported to commonly induce hallucinations, nightmares, and delusions, which led to its nonuse in medicine (White & Roth 2012; Coursey 1978).
Are dysdelics always dysphoric?
This is unlikely. There are already cases where individuals report neutral or positive effects during Salvia use. Even the dysphoric effects of dynorphin seem to require a co-factor known as corticopterin-releasing hormone (CRH) (Land et al 2008). The dysphoric effect only seems to occur when both of these stress neurotransmitters are simultaneously active in the brain. Most people may experience dysphoria on dysdelics because they anticipate dysphoria or they fear the drug or its’ effects. The fear and stress of taking the new drug may commonly produce a CRH response that aides the dysdelic in producing a dysphoric reaction. Novel experiences stimulate CRH activity in animals (Zarrow, Campbell, & Denenberg 1972) (although context seems to matter here), so perhaps most people trying salvia will have the dual conditions necessary for aversive effects.
Perhaps the kind of novelty-seekers who try many novel drugs, such as dysdelics, are more likely to have a positive reaction to salvia, since they chase novel experiences despite CRH activity, suggesting they might not have aversion. Not to mention, Salvia and other dysdelics are pretty far down the list of most people’s ‘must-try’ drugs. If anyone is going to try out the dysdelics, it is probably largely the novelty-seekers and the ones who are naïve about the drug’s effects.
Another important bit: 2 distinct subpopulations of dynorphin neurons in the nucleus accumbens drive aversion and reward (Al-Hasani et al., 2015). In other words, there are different kinds of dynorphin neurons that produce opposite effects, reward and aversion, which could be a factor in whether the drug produces dysphoria or not. Though, it is clear that aversive reactions are common with KOR agonists.
The term dysdelic may not encompass every possible phenomenological effect that KOR agonists produce, but most drug class names also do not. Psychedelics are named so for their ability to produce insight into our selves, but this isn’t necessarily the main effect nor is it guaranteed to occur. Many drugs are capable of producing paradoxical effects, which essentially means they have broken the stereotype of their drug class. Rather than talking about these drugs as being psychedelics or dysdelics, it may be better to say that a drug is capable of eliciting psychedelic or dysdelic phenomenologies. This goes for any drug class where the drug class is named after its’ subjective effects (stimulants, sedatives, dissociatives, psychedelics, dysdelics). The dysdelic state may be most commonly elicited by the KOR agonists, even if it is not the guaranteed type of effect. These naming conventions given to drug classes are not necessarily reflective of the most scientific naming, but it is an artform that characterizes the kind of stories we tell about the various types of drugs and that is something beautiful.
Subjective Effects
The dysdelia experience is often characterized by dysphoria, anxiety, dissociation, out-of-body experiences, total body physical pain, stretching and distorting of the body’s form, and immersion into strange and often novel universes. The subjective effects described here will mostly focus on salvia, the most commonly used dysdelic.
Some users report spending years in the salvia dysdelic space, despite that salvia lasts only a short period of time, often mere minutes. One could only imagine the absurd effects that could occur with high doses of longer lasting dysdelics. Whether or not these long-lasting subjective experiences are purely a myth is uncertain, but there are at least multiple reports of such extreme ‘time-dilation’ (interesting Reddit anecdotes: 1, 2). This sense of an eternally lasting hellscape may not come down to subjectively experiencing of eternal amounts of time. Consider that we only really experience time in the present moment and our sense of how long something has been happening is something that we sense only in the present moment too. We may have a specific feeling about how much time has passed. Like the memory for 5 minutes ago could appear faint and ancient as if much time has passed. Salvia likely doesn’t produce the sense of eternity by actually causing eternal experiences, but only an illusion of such.
The dissociation produced by dysdelics resembles the dissociative drugs, though not entirely. The effect seems to be somehow partial compared to the classic dissociatives, where more extreme distortions of perception occur at even lower levels of body intoxication. One tends to be very ‘drunk’ on dissociatives before anything like an alternate universe appears, while salvia may produce trips more potently without as many tangential effects. Dysdelics like salvia extract also produce an extreme launch-style effect that hurls you into another world. This other world may be comparable to the k-hole that ketamine and other dissociatives produce.

Besides an induced dreaming (nightmare) effect, there are external hallucinations reported on dysdelics. One study injected the body’s own dysdelic, dynorphin, to heroin addicts in hopes that it may help in treating addiction and the researchers found that 1/4 of the participants had hallucinations of insects crawling on their body (Wen & Ho 1982).
Experiences
In my own experience, when a plain leaf of salvia is vaporized, the effect seems to induce 2-dimensionality of perception, shiny geometric sparkles on the walls, and dissociative amnesiac type effects. The effect has sometimes left me feeling traumatized after, even if the experience wasn’t really scary. There was a recovery time before I could access my fully functioning mind again. Sometimes the onset of effects felt as if the spirit world was ripping open and I was beginning to see the underlay of reality, the secret behind the screen that I am used to seeing in daily life.
This seems to resemble one of the low dose salvia trip reports listed on the PsychonautWiki:
I held the smoke in for about 5 seconds then exhaled, which elicited gasps of dismay from two friends.
“You’re supposed to hold it in man, now it probably won’t work”, they advised. However, within a few seconds, I could see things beginning to change; my friends’ faces looked like some kind of blurry plasticine. I sunk into the couch and clutched its armrest with anticipation. They could see my expression changing quickly, and remarked that I was obviously starting to trip. I looked at the TV across the room and was shocked that I could see the entire TV. It was like I could see all six sides of the TV unfolded before me, including its wiry innards. My mouth gaped as I took all of this in.
From: a tall humanoid figure wearing a white cloak
The same person took a second dose shortly after and things got quite a bit more intense:
I held the acrid smoke in and saw the same plasticine-face effect as my friends looked at me. But this time there was a very strong rush. It felt ominous and there seemed to be a “woosh” sound, like when one puts one’s ear to a seashell. I could tell this was going to be intense, and once again I laid back and clutched the couch. Then blackness.
I opened my eyes, to see that I was standing on the edge of a plateau. The ground beneath me was powdery and orange-red, like Mars. The clear sky was a similar color, but more reddish. There were wisps of white clouds, but not many. I took a few steps toward the edge of the cliff. Far below me was a vast orange desert-badlands sort of area. The ground was cracked and dry. I heard the woosh again and looked upward; several creatures were flying overhead. They were shaped like manta rays with very long dragon-like tails. They were dark shades of purple and blue, with bumpy leathery scales. I spoke their name, which to the best of my recollection was something like “Quixocitolix” (no, not Quetzalcoatl). It was as though I had seen these creatures before. They did not seem dangerous, but they were worthy of my respect and appreciation.
From: a tall humanoid figure wearing a white cloak

Dissociation and Visuals
Historically Salvia has been lumped in with the dissociative drugs like ketamine, PCP, DXM, and MXE. The mechanism of dissociative effects for the classic dissociative drugs involves shutting down glutamate receptors known as NMDA receptors. Salvia induces dissociative effects through its’ KOR agonism, which does seem to impact glutamate activity (Hjelmstad & Fields 2003; Manuef et al., 1995; Werkheiser et al., 2007; Wagner, Terman, & Chavkin 1993), likely explaining these dissociative effects. There are a few ways the dysdelics differ from classic dissociative drugs.
There are some overlaps in mechanisms between dissociatives and KOR agonists. KOR agonism reduces glutamate release, possibly decreasing both AMPA receptor and NMDA receptor activity, while dissociatives reduce only NMDA receptor activity and may even indirectly enhance AMPA receptor activity through glutamate disinhibition (Lorrain et al 2003). The effect of KOR agonism on glutamate activity is likely to be selective to KOR containing neurons or peripherally related systems, while classic dissociatives may be fairly indiscriminate, targeting any neuron with NMDA receptors. Hypothetically, this could be noted in the difference in subjective effects observed by both classes of drugs, as dissociatives produce much more extreme drunkeness before hallucinations and weird experiences start to emerge, whereas the dysdelic Salvia produces body numbness and balance changes too, but the mind changes are much more drastic and occur earlier in the body intoxication-spectrum than compared with dissociatives (though, this could be due to method of ingestion, smoking versus oral/insufflation). Even though the effect of dissociation feels partial, this partiality seems to allow the user to dive much further into dissociative mindscapes than even the classic dissociatives themselves.
Becoming Objects
This is a rather peculiar yet commonly reported effect of salvia. Users of salvia often report that they have become some object in their visual field or their dreamscape. It is often that the user ‘becomes’ a couch, becoming a wall, becoming a vase, and so on. This is one of the more peculiar effects of the drug and might be a form of dissociation combined with amnesia that leads one to forget what they are and what they should expect themselves to be. This may lead the user to assume that they are the thing that they are observing, often times an inanimate object.
AIWS
One of the interesting studies on dysdelic states compared the phenomenology of the experience to Alice In Wonderland Syndrome (AIWS). This syndrome usually involves perceptual alterations to one’s sense of object size, distance, and other peculiar distortions. Sometimes patients report that objects appear massive or tiny, close up or very far away.
A paper (not peer-reviewed) (Hesselink 2019) has compared the phenomenological experiences discovered under salvia divinorum to the ones observed in those with AIWS. Notably, the author’s explored alterations of body representation involving tactile feedback that seems to exist in both disorders. Some of the cases they present of subjects mention strange phenomenological experiences like their body being sucked into a spiral at their right side or merging with the carpet.
Dysdelics As Cognitive Atomizers
I’ve described the effects of NMDAr reducing drugs in Desummation and Cognitive Atomization. I think that these arguments also may explain the effects of dysdelics. My argument is that conditioned memory frameworks guide the way we perceive the world, the way we think about the world, our behaviors and our ability to navigate the external world. The NMDAr activity reducing drugs may reduce access to these guiding frameworks and influence them on a spectrum that is dose-dependent. Lower doses may cut our ability to encode new memories, while higher doses may shut down the ability to guide our world models through our historic mental references, in other words, everything that we have learned may be disrupted, causing us to be unfamiliar with the external world and even our own bodies.
In these projects I’ve described an effect I’ve termed cognitive atomization, which refers to a disruption in the brain’s coincidence detection and possibly previously encoded coincidence-based frameworks of cognition. This can cause the structures forming our cohesive and connected experience to fragment, to atomize. One way we can observe this with dissociative drugs that they inhibit the ability to see optical illusions (Jacobsen, Barros, & Maior, 2017). This also seems to occur in THC users (Koethe, Gerth, Neatby et al 2006). Illusions are kind of like a perceptual auto-correct in the brain. Similar to text-correction and predictive text on your phone, the perceptual auto-correct is sometimes wrong as well. Optical illusions are like those errors, except with perception.
But everything is an illusion. At least this is what is argued in Desummation. The way that we observe an object to exist as a cohesive symbol or structure, as opposed to atomized bits of disconnected pixels is an illusion. So on higher doses of the dissociatives, we see something that appears to be a lot like apperceptive agnosia, which is the inability to recognize objects. Salvia seems to share these properties with dissociatives, though most trip reports you will find involve higher doses that entirely cut you off from external reality, at which point the experience is different.
A comparison of apperceptive agnosia and associative agnosia.
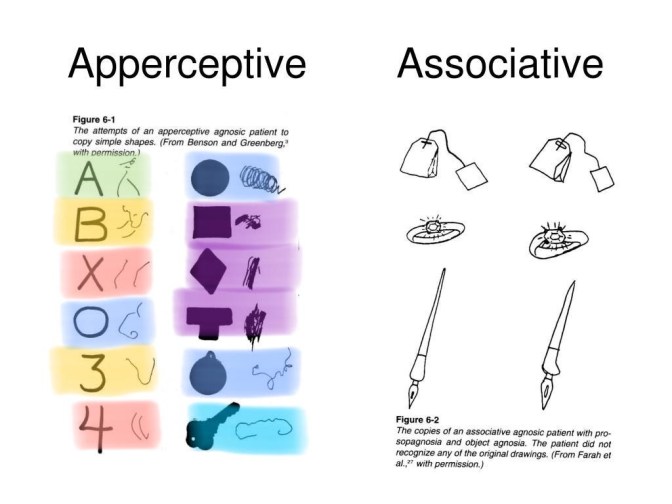
Once these drugs cut one off from external experience altogether, the experience is likely much more akin to dreaming. This is something I cannot yet explain through these chemical models.
Lower doses of atomizing drugs seem to frequently induce a higher state of consciousness and perceptual clarity. This may be due to the way we develop learned perceptual filters to help us reduce the amount of sensory information that we normally experience and minimize the sensory overload of daily life. The low dose of an atomizing drug may induce a level of unfamiliarity that we are, well, familiar with in adult life. Low-grade novel experiences such as moving to a new home seem to induce a kind of raised state of consciousness, as we are suddenly without our cozy habituated lifestyles, living outside of our comfort bubbles.
As the dose of an atomizer increases, one will even lose the sense of familiarity with their own body, their own mind, and their perceptual frameworks that maintain their mental structures based on learned patterns of the world. One may begin to see new visual patterns as their brain attempts to forge meaning in the chaos of sudden unfamiliarity.
At increasingly higher doses of atomizing drugs, one may experience the creeping emergence of the void. As coincidence detection and recognition begins to fail ubiquitously, only distinct pixels of qualia are experienced, pixels that do not connect to form meaning. Beyond this, exists a void of meaning and qualia. The final stage of this progressive elimination of qualia may be unconsciousness, the kind of state that people utilize during surgery, an anesthetic ‘coma’.
People also report out of body experiences on dysdelics.

Bodily Sensations
The bodily experience of dysdelia ranges from totally leaving the body and experiencing total immense suffering, ripping, compressing, and morphing of the body. It is sometimes reported that people sense gravity or ripping of their body as their dose of salvia kicks in. This is something that isn’t really reported on other drugs as far as I know.
Gravitational alterations are often reported from the use of Salvia. From a study (Stiefel, Merrifield, & Holcombe 2014) that compiled the reported effects of Salvia:
A fourth aspect of experience for which Salvia scored significantly higher was the “non-visual sensory” category, with the difference mainly driven by the following questionnaire items: “Gravity was in the wrong direction, or wrong force field,” “Feel as moving/falling/flying through space,” as well as the non-specific “Body feels different”.
Bodily sensations known as formication have been described in human studies involving injections of dynorphin (Wen & Ho 1982). This sensation is one of insects crawling on the skin. In the past, I’ve tied the KOR system to these bodily sensations in the post Illusory Bugs.
I tested using salvia daily in small doses, for a seeming antidepressant rebound effect that occurred after the acute effects disappear. After about a week, these benefits ceased, something that mirrors chronic ketamine use as well (Li et al., 2017; Fan et al., 2016). After this point, I began to experience mild psychotic, dissociative, and numbing effects that persisted even beyond the acute effects. At some point, I developed formication, the sensation of crawling insects on my body.
This formication would return upon stress, consumption of cannabis, and during the comedown of other substances that stimulate dopamine activity. I ended up writing about this experience and potential solutions in the post Illusory Bugs.
Distinctions From Other Hallucinogens
Psychedelics, dissociatives, and deliriants are the most commonly talked about hallucinogenic drugs. Deliriant drugs seem to function more differently from all the hallucinogen types mentioned here, so these won’t be explored as much here. The deliriants are often sedative drugs that induces waking dream-like states and amnesia. Usually producing real hallucinations, in the sense that you are in the normal world, but seamless hallucinatory experiences keep occurring that you are unable to distinguish from real experiences.
Dissociatives (Lorrain et al. 2003), psychedelics (Moreno et al. 2013), and dysdelics (Hjelmstad & Fields, 2003; Manuef et al., 1995; Werkheiser et al., 2007) all modulate glutamate function in the brain, though in distinct ways. This glutamatergic modulation may underlie their strange effects on perception and cognition.
The dissociatives and anticholinergics seem to work by shutting down the normal function of neurotransmitters in the brain. On the other hand, psychedelics and dysdelics are unique in that they produce their effects by mimicking endogenous neurotransmitters, in the agonist direction. They both enhance function of a dedicated subjective experience system of the brain. For some reason, it seems we are built to experience effects along the salvia-space. Meanwhile, dissociative drugs function by disrupting a normal function of a common glutamate receptor, the NMDAr. So now we have at least two receptor system types, serotonin and dynorphin, that produce radical shifts in conscious experience and they both appear to work through directly interacting opposite mechanisms. It is worth mentioning that this characterization could be totally arbitrary, as both agonism and antagonism could be viewed as disrupting normal function and there are even endogenous receptor antagonists, dynorphin being an NMDA receptor antagonist itself.
There has been at least one study comparing the subjective experience of dysdelics and psychedelics (Maqueda et al., 2015). This study noted differences like that the dysdelic drug produced auditory hallucinations and loss of body contact more frequently than the psychedelic drugs.
From that study:
The inhalation of vaporized salvinorin-A led to very strong psychotropic effects of rapid onset and short duration. Perceptual modifications included the visual domain, and in contrast with 5HT2A agonists, auditory hallucinations were very common. Also in contrast with the classical serotonergic psychedelics, loss of contact with external reality was prominent with the participants being unreactive to external visual and verbal cues, especially after the medium and high doses. While at the low and medium doses there was an increase in bodily sensations, at 1.0mg there was an almost complete loss of body ownership and an increase in out-of-body experiences. These results suggest that the dynorphins – KOR system may play a previously underestimated role in the regulation of sensory perception, interoception, and the sense of body ownership in humans.
Dysdelic Theory

Follow Utheraptor’s Art!
Dysdelics may be anti-psychedelic.
(and psychotic)
Keep in mind, this section gets increasingly obscure and speculative.
Dysdelics can be viewed as anti-psychedelics. Besides seeming like the antithesis of the classic psychedelic drugs, the mechanisms that underlie the effects of dysdelics actually may be somewhat opposite as well. Psychedelics work by stimulating serotonergic mechanisms, usually 5HT2a, 5HT2c and 5HT1a receptors. On the other hand, dysdelics operate by stimulating dynorphinergic mechanisms, namely the KORs. The serotonergic and dynorphinergic systems interact in interesting and contrary ways that support this notion that dysdelics are anti-psychedelic. Though this may be a simplification, it is still interesting and potentially useful to note.
5HT1a and 5HT2a receptors both seem to have mechanisms that suppress KOR activity. A 5HT1a receptor stimulating drug was found to prevent the increase of dynorphin activity that usually occurs when the dopaminergic drug L-DOPA is applied in animals (Tomiyama et al, 2005). 5HT2a receptor stimulation is shown to increase acetylcholine in the prefrontal cortex and hippocampus of rats (Nair & Gudelsky, 2004), which is notable because one of the cholinergic receptors known as nAch alpha7 seems to suppress dynorphin release from microglia (Ji et al., 2019). The serotonergic psychedelic, LSD, suppresses the depressant effects of dynorphin/KOR activity (Sakloth et al., 2019), which may occur through 5HT2ar-mGlur2 complexes in which 5HT2a receptor agonism leads to suppression of mGlu2 receptor activity (dos Santos, 2014), since mGlu2 receptors may facilitate dynorphin activity (Liu et al., 2017). This mGlu2 explanation is complex and hard to parse, especially since the only study I found exploring mGlu2 and dynorphin was looking at the estrus cycle, pain sensitivity, and focused on mGlu1 and connections that mGlu2/3 have to mGlu1. In support of 5HT1a- and 5HT2a-dependent dynorphin suppressing mechanisms, various studies have shown that 5HT1a and 5HT2a receptor stimulation suppresses aversion processing (Melo & Brandão, 1995; Nogueira & Graeff, 1995; Kuypers et al., 2018; de Paula Soares et al., 2010) and dynorphin is implicated in aversion processing (Massaly et al 2019; Land et al 2008).
On the other hand, ketanserin, a decently selective 5HT2a receptor antagonist has been found to potentiate the KOR agonist effects of morphine (Peiró et al., 2011) and has been shown not to disrupt the dysdelic effects of Salvia (Maqueda et al., 2016). Morphine also seems to decrease 5HT2a receptor binding (Adriaens et al., 2010), though it is unclear if this mechanism is KOR-dependent or maybe even MOR-dependent. The withdrawals of morphine may involve 5HT2a receptor activity as well (Pang et al., 2016). These suggest that some antagonistic relationship between 5HT2a receptors and KORs may exist.
Another link between the dysdelics and the serotonin system is at the serotonin transporter. KOR stimulation invokes the serotonin transporter, which removes serotonin so that it doesn’t bind to the serotonin receptors. This removal of serotonin seems to be necessary for the aversive effects of KOR stimulation (Schindler et al., 2012). Perhaps this is because serotonin 5HT2a and 5HT1a receptors suppress aversion and the serotonin transporter activation can prevent this from occurring by removing serotonin, thus facilitating aversion by disinhibiting dynorphin further. The effect of KOR in this study also revealed that the removal of serotonin may enhance cocaine reward. So in this sense, dysdelics may enhance the rewarding effects of other drugs. On the opposite end, SSRI drugs diminish reward and aversion processing by blocking serotonin transporter function, which increases serotonin available to bind on serotonin receptors (McCabe et al. 2010).
This suggests that dysdelics (KOR agonists) and psychedelics (serotonin agonists) may have very opposing patterns of effects. While SSRI drugs suppress both reward and aversion processing, psychedelics don’t exactly seem to do this. Instead, people often view psychedelics as having high variance in valence, meaning that the emotional effects of the drug can vary widely, from good to bad. Though, SSRI drugs aren’t entirely numbing to affect. In fact, the initial effects of these drugs often produce anxiety, which is thought to be mediated by one of the common psychedelic receptor targets, 5HT2c (Pelrine et al., 2016; Bagdy et al., 2001; Burghardt et al., 2007). This receptor type is associated with PTSD (Règue et al., 2019), CRH release (Heisler et al., 2007), and anxiety (Harada, Yamaji, & Matsuoka, 2008), which are all associated with dynorphin activity too. Besides being a co-factor with dynorphin in the negative emotional feeling produced by stress, CRH is observed to actually increase dynorphin release (Bruchas, Land, Lemos, & Chavkin, 2009; Nikolarakis, Almeida, & Herz, 1986), which probably functions in some cascade of the stress response. So it is possible that negative valence psychedelic experiences are actually KOR-mediated like salvia, but through the 5HT2c receptor rather than KOR binding directly.
The 5HT1a receptor is another common target of psychedelic drugs and is thought to underlie one of the main antidepressant effects of serotonergic drugs, inducing plasticity and neurogenesis (Albert & Vahid-Ansari, 2019). This receptor type releases endorphins (MOR agonists) (Navinés et al., 2008), which might explain positive valence effects of psychedelics. As previously mentioned, this receptor also seems to suppress the release of dynorphin that occurs via dopaminergic drugs (Tomiyama et al, 2005).
Serotonin and dynorphin may be antagonistic.
(although this is definitely an oversimplification, it might still be a useful model)
This paints an interesting picture on the effects of both the dysdelic and psychedelic mechanisms in the brain. Though it is important to note that this relationship clearly isn’t simple enough to say that psychedelics are always positive valence or that dysdelics are always negative valence. Though there does seem to be a general trend in those directions. We also cannot conclude that salvia and and psychedelics antagonize each other. This relationship between dysdelics and psychedelics isn’t perfect.
It is also possible that high doses of psychedelics may bring on dysdelic responses by rapidly desensitizing or downregulating the 5HT2a receptor. The come up may be “psychedelic” while after the peak it may turn suddenly dysdelic as 5HT2a receptor activity starts decreasing from peak levels. This would resemble the kind of pattern seen in seizures, where initially a spark of increased consciousness and sensitivity may emerge from a storm of glutamate, but then dynorphins flood our system and produce dissociative and psychotic-like effects from the sudden crash and attenuation of glutamate signaling (Bortolato & Solbrig, 2007; Loacker et al., 2007). In states of peak consciousness, our brain may violently push back in order to maintain homeostasis, and then once these mechanisms inducing the peak begin to fade, we may find ourselves hypo-conscious until yet again homeostatic mechanisms come into play to bring us into balance. So these different states may not be totally exclusive to each respective drug, though I feel we should categorize these states based on the agonist effect of these receptors rather than homeostatic responses induced by the receptor activity.
Dysdelic Therapy
Since ketamine seems to exert some of its’ antidepressant effects by stimulating KOR and desensitizing the system, the same may be true of other KOR agonists. Ketamine may be a dysdelic itself, but it has other major hallucinogenic mechanisms itself so it is not a good representative of dysdelic drugs. This opens up the possibility that dysdelics could help by essentially smashing on the ‘mental illness’ systems of the brain so that afterwards one is left desensitized to aversion, psychotic symptoms, depression, anxiety, stress and maybe even pain.
Frequent low doses of dysdelics might work to balance mood and reduce mania, seemingly without even producing their usual psychotomimetic effects (Cohen & Murphy, 2008). Mania reduction makes sense, especially if the serotonin transporter really does upregulate and ‘reverse tolerance’ sets in. The risk would be that KOR is desensitized and antidepressant-like manic effects come into play.
. . .
If you found this enjoyable, consider joining the Patreon! I’ve been posting detailed experience reports with my adventures using prescription ketamine. Also. someone sent me an EEG device to collect data on ketamine-induced brainwave changes which I’ve started posting there too. I also post secret mini podcasts. You can find the publicly available podcasts here by the way!
Special thanks to the 14 patrons: Buttercup, Libby Rosebaugh, Dan Elton, Idan Solon, David Chang, Jack Wang, Richard Kemp, Alex W, Sarah Gehrke, Melissa Bradley, Morgan Catha, Niklas Kokkola, Riley Fitzpatrick, and Charles Wright! Abhi is also the artist who created the cover image for Most Relevant. Please support him on instagram, he is an amazing artist! I’d also like to thank Alexey Guzey, Annie Vu, Chris Byrd, and Kettner Griswold for your kindness and for making these projects and the podcast possible through your donations.
If you’d like to support these projects like this, check out this page.
If you liked this, follow me on

Citations
Adriaens, A., Vermeire, S., Waelbers, T., Duchateau, L., Sys, S., Dobbeleir, A., … & Peremans, K. (2010). Morphine decreases 5-HT2A receptor binding measured with SPECT in the canine frontal cortex. In 23rd Annual congress of the European Association of Nuclear Medicine (EANM 2010) (Vol. 37, No. suppl. 2, pp. S389-S389).
Al-Hasani, R., McCall, J. G., Shin, G., Gomez, A. M., Schmitz, G. P., Bernardi, J. M., … & Bruchas, M. R. (2015). Distinct subpopulations of nucleus accumbens dynorphin neurons drive aversion and reward. Neuron, 87(5), 1063-1077.
Albert, P. R., & Vahid-Ansari, F. (2019). The 5-HT1A receptor: signaling to behavior. Biochimie, 161, 34-45.
Bagdy, G., Graf, M., Anheuer, Z. E., Modos, E. A., & Kantor, S. (2001). Anxiety-like effects induced by acute fluoxetine, sertraline or m-CPP treatment are reversed by pretreatment with the 5-HT2C receptor antagonist SB-242084 but not the 5-HT1A receptor antagonist WAY-100635. International Journal of Neuropsychopharmacology, 4(4), 399-408.
Bilkei-Gorzo, A., Erk, S., Schürmann, B., Mauer, D., Michel, K., Boecker, H., … & Zimmer, A. (2012). Dynorphins regulate fear memory: from mice to men. Journal of Neuroscience, 32(27), 9335-9343.
Bortolato, M., & Solbrig, M. V. (2007). The price of seizure control: dynorphins in interictal and postictal psychosis. Psychiatry research, 151(1-2), 139-143.
Bruchas, M. R., Land, B. B., Lemos, J. C., & Chavkin, C. (2009). CRF1-R activation of the dynorphin/kappa opioid system in the mouse basolateral amygdala mediates anxiety-like behavior. PloS one, 4(12), e8528.
Burghardt, N. S., Bush, D. E., McEwen, B. S., & LeDoux, J. E. (2007). Acute selective serotonin reuptake inhibitors increase conditioned fear expression: blockade with a 5-HT2C receptor antagonist. Biological psychiatry, 62(10), 1111-1118.
Clark, S. D., & Abi-Dargham, A. (2019). The role of dynorphin and the kappa opioid receptor in the symptomatology of schizophrenia: A review of the evidence. Biological psychiatry, 86(7), 502-511.
Cohen, B. M., & Murphy, B. (2008). The effects of pentazocine, a kappa agonist, in patients with mania. International Journal of Neuropsychopharmacology, 11(2), 243-247.
Coursey, C. E. (1978). The psychotomimetic side effects of pentazocine. Drug Intelligence & Clinical Pharmacy, 12(6), 341-346.
Crowley, N. A., Bloodgood, D. W., Hardaway, J. A., Kendra, A. M., McCall, J. G., Al-Hasani, R., … & Lowell, B. B. (2016). Dynorphin controls the gain of an amygdalar anxiety circuit. Cell reports, 14(12), 2774-2783.
de Paula Soares, V., Campos, A. C., de Bortoli, V. C., Zangrossi Jr, H., Guimarães, F. S., & Zuardi, A. W. (2010). Intra-dorsal periaqueductal gray administration of cannabidiol blocks panic-like response by activating 5-HT1A receptors. Behavioural brain research, 213(2), 225-229.
dos Santos, R. G. (2014). Psychedelics, Glutamate, and Neuroimaging Studies. Anesthesiology, 120(6), 1521-1522.
Fan, N., Xu, K., Ning, Y., Rosenheck, R., Wang, D., Ke, X., … & He, H. (2016). Profiling the psychotic, depressive and anxiety symptoms in chronic ketamine users. Psychiatry research, 237, 311-315.
Gold, M. S., Blum, K., Febo, M., Baron, D., Modestino, E. J., Elman, I., & Badgaiyan, R. D. (2018). Molecular role of dopamine in anhedonia linked to reward deficiency syndrome (RDS) and anti-reward systems. Front Biosci (Schol Ed), 10, 309-325.
Harada, K., Yamaji, T., & Matsuoka, N. (2008). Activation of the serotonin 5-HT2C receptor is involved in the enhanced anxiety in rats after single-prolonged stress. Pharmacology Biochemistry and Behavior, 89(1), 11-16.
Heisler, L. K., Zhou, L., Bajwa, P., Hsu, J., & Tecott, L. H. (2007). Serotonin 5‐HT2C receptors regulate anxiety‐like behavior. Genes, Brain and Behavior, 6(5), 491-496.
Hesselink, J. M. K. Salvia divinorum induces body awareness changes comparable to metamorphic symptoms in the Alice in Wonderland syndrome.
Hjelmstad, G. O., & Fields, H. L. (2003). Kappa opioid receptor activation in the nucleus accumbens inhibits glutamate and GABA release through different mechanisms. Journal of neurophysiology, 89(5), 2389-2395.
Jacobsen, M. E., Barros, M., & Maior, R. S. (2017). MK-801 reduces sensitivity to Müller-Lyer’s illusion in capuchin monkeys. Behavioural Brain Research, 316, 54-58.
Ji, L., Chen, Y., Wei, H., Feng, H., Chang, R., Yu, D., … & Zhang, M. (2019). Activation of alpha7 acetylcholine receptors reduces neuropathic pain by decreasing dynorphin A release from microglia. Brain research, 1715, 57-65.
Knoll, A. T., & Carlezon Jr, W. A. (2010). Dynorphin, stress, and depression. Brain research, 1314, 56-73.
Koethe, D., Gerth, C. W., Neatby, M. A., Haensel, A., Thies, M., Schneider, U., … & Leweke, F. M. (2006). Disturbances of visual information processing in early states of psychosis and experimental delta-9-tetrahydrocannabinol altered states of consciousness. Schizophrenia research, 88(1-3), 142-150.
Kuypers, K. P., De La Torre, R., Farre, M., Pizarro, N., Xicota, L., & Ramaekers, J. G. (2018). MDMA-induced indifference to negative sounds is mediated by the 5-HT 2A receptor. Psychopharmacology, 235(2), 481-490.
Land, B. B., Bruchas, M. R., Lemos, J. C., Xu, M., Melief, E. J., & Chavkin, C. (2008). The dysphoric component of stress is encoded by activation of the dynorphin κ-opioid system. Journal of Neuroscience, 28(2), 407-414.
Li, C. S. R., Zhang, S., Hung, C. C., Chen, C. M., Duann, J. R., Lin, C. P., & Lee, T. S. H. (2017). Depression in chronic ketamine users: sex differences and neural bases. Psychiatry Research: Neuroimaging, 269, 1-8.
Liu, N. J., Murugaiyan, V., Storman, E. M., Schnell, S. A., Kumar, A., Wessendorf, M. W., & Gintzler, A. R. (2017). Plasticity of signaling by spinal estrogen receptor alpha, kappa-opioid receptor and mGluRs over the rat reproductive cycle regulates spinal endomorphin 2 antinociception: relevance of endogenous biased agonism. Journal of Neuroscience, 1927-17.
Loacker, S., Sayyah, M., Wittmann, W., Herzog, H., & Schwarzer, C. (2007). Endogenous dynorphin in epileptogenesis and epilepsy: anticonvulsant net effect via kappa opioid receptors. Brain, 130(4), 1017-1028.
Lorrain, D., Baccei, C. S., Bristow, L. J., Anderson, J. J., & Varney, M. A. (2003). Effects of ketamine and N-methyl-D-aspartate on glutamate and dopamine release in the rat prefrontal cortex: modulation by a group II selective metabotropic glutamate receptor agonist LY379268. Neuroscience, 117(3), 697-706.
Maneuf, Y. P., Mitchell, I. J., Crossman, A. R., Woodruff, G. N., & Brotchie, J. M. (1995). Functional implications of kappa opioid receptor-mediated modulation of glutamate transmission in the output regions of the basal ganglia in rodent and primate models of Parkinson’s disease. Brain research, 683(1), 102-108.
Maqueda, A. E., Valle, M., Addy, P. H., Antonijoan, R. M., Puntes, M., Coimbra, J., … & Riba, J. (2016). Naltrexone but not ketanserin antagonizes the subjective, cardiovascular, and neuroendocrine effects of salvinorin-A in humans. International Journal of Neuropsychopharmacology, 19(7).
Maqueda, A. E., Valle, M., Addy, P. H., Antonijoan, R. M., Puntes, M., Coimbra, J., … & Riba, J. (2015). Salvinorin-A induces intense dissociative effects, blocking external sensory perception and modulating interoception and sense of body ownership in humans. International Journal of Neuropsychopharmacology, 18(12), pyv065.
Massaly, N., Copits, B. A., Wilson-Poe, A. R., Hipólito, L., Markovic, T., Yoon, H. J., … & Klaas, A. (2019). Pain-induced negative affect is mediated via recruitment of the nucleus accumbens kappa opioid system. Neuron, 102(3), 564-573.
McCabe, C., Mishor, Z., Cowen, P. J., & Harmer, C. J. (2010). Diminished neural processing of aversive and rewarding stimuli during selective serotonin reuptake inhibitor treatment. Biological psychiatry, 67(5), 439-445.
Melo, L. L., & Brandão, M. L. (1995). Role of 5-HT1A and 5-HT2 receptors in the aversion induced by electrical stimulation of inferior colliculus. Pharmacology Biochemistry and Behavior, 51(2-3), 317-321.
Moreno, J. L., Holloway, T., Albizu, L., Sealfon, S. C., & González-Maeso, J. (2011). Metabotropic glutamate mGlu2 receptor is necessary for the pharmacological and behavioral effects induced by hallucinogenic 5-HT2A receptor agonists. Neuroscience letters, 493(3), 76-79.
Nair, S. G., & Gudelsky, G. A. (2004). Activation of 5‐HT2 receptors enhances the release of acetylcholine in the prefrontal cortex and hippocampus of the rat. Synapse, 53(4), 202-207.
Navinés, R., Martín-Santos, R., Gómez-Gil, E., de Osaba, M. J. M., & Gastó, C. (2008). Interaction between serotonin 5-HT1A receptors and β-endorphins modulates antidepressant response. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 32(8), 1804-1809.
Nikolarakis, K. E., Almeida, O. F. X., & Herz, A. (1986). Stimulation of hypothalamic β-endorphin and dynorphin release by corticotropin-releasing factor (in vitro). Brain research, 399(1), 152-155.
Nogueira, R. L., & Graeff, F. G. (1995). Role of 5-HT receptor subtypes in the modulation of dorsal periaqueductal gray generated aversion. Pharmacology Biochemistry and Behavior, 52(1), 1-6.
Pang, G., Wu, X., Tao, X., Mao, R., Liu, X., Zhang, Y. M., … & Zhang, G. (2016). Blockade of serotonin 5-HT2A receptors suppresses behavioral sensitization and naloxone-precipitated withdrawal symptoms in morphine-treated mice. Frontiers in pharmacology, 7, 514.
Peiró, A. M., Climent, L., Zapater, P., Horga, A., & Horga, J. F. (2011). Ketanserin potentiates morphine-induced antinociception mediated by kappa-receptor activation. Pharmacological research, 64(1), 80-84.
Pelrine, E., Pasik, S. D., Bayat, L., Goldschmiedt, D., & Bauer, E. P. (2016). 5-HT2C receptors in the BNST are necessary for the enhancement of fear learning by selective serotonin reuptake inhibitors. Neurobiology of learning and memory, 136, 189-195.
Rabellino, D., Densmore, M., Harricharan, S., Jean, T., McKinnon, M. C., & Lanius, R. A. (2018). Resting‐state functional connectivity of the bed nucleus of the stria terminalis in post‐traumatic stress disorder and its dissociative subtype. Human Brain Mapping, 39(3), 1367-1379.
Règue, M., Poilbout, C., Martin, V., Franc, B., Lanfumey, L., & Mongeau, R. (2019). Increased 5-HT2C receptor editing predisposes to PTSD-like behaviors and alters BDNF and cytokines signaling. Translational psychiatry, 9(1), 1-15.
Sakloth, F., Leggett, E., Moerke, M. J., Townsend, E. A., Banks, M. L., & Negus, S. S. (2019). Effects of acute and repeated treatment with serotonin 5-HT2A receptor agonist hallucinogens on intracranial self-stimulation in rats. Experimental and clinical psychopharmacology, 27(3), 215.
Schindler, A. G., Messinger, D. I., Smith, J. S., Shankar, H., Gustin, R. M., Schattauer, S. S., … & Chavkin, C. (2012). Stress produces aversion and potentiates cocaine reward by releasing endogenous dynorphins in the ventral striatum to locally stimulate serotonin reuptake. Journal of Neuroscience, 32(49), 17582-17596.
Stiefel, K. M., Merrifield, A., & Holcombe, A. O. (2014). The claustrum’s proposed role in consciousness is supported by the effect and target localization of Salvia divinorum. Frontiers in integrative neuroscience, 8, 20.
Tomiyama, M., Kimura, T., Maeda, T., Kannari, K., Matsunaga, M., & Baba, M. (2005). A serotonin 5-HT1A receptor agonist prevents behavioral sensitization to L-DOPA in a rodent model of Parkinson’s disease. Neuroscience research, 52(2), 185-194.
Wagner, J. J., Terman, G. W., & Chavkin, C. (1993). Endogenous dynorphins inhibit excitatory neurotransmission and block LTP induction in the hippocampus. Nature, 363(6428), 451-454.
Wen, H. L., & Ho, W. K. (1982). Suppression of withdrawal symptoms by dynorphin in heroin addicts. European journal of pharmacology, 82(3-4), 183-186.
Werkheiser, J. L., Rawls, S. M., & Cowan, A. (2007). Nalfurafine, the kappa opioid agonist, inhibits icilin-induced wet-dog shakes in rats and antagonizes glutamate release in the dorsal striatum. Neuropharmacology, 52(3), 925-930.
White, K. L., & Roth, B. L. (2012). Psychotomimetic effects of kappa opioid receptor agonists. Biological psychiatry, 72(10), 797-798.
Zarrow, M. X., Campbell, P. S., & Denenberg, V. H. (1972). Handling in infancy: increased levels of the hypothalamic corticotropin releasing factor (CRF) following exposure to a novel situation. Proceedings of the Society for Experimental Biology and Medicine, 141(1), 356-358.
Two relevant talks by a University of Toronto professor:
LikeLike