ABSTRACT
This research proposal aims to test the hypothesis that NMDA and AMPA glutamate receptors are differentially involved in temporal acuity of visual perception, which is in essence, the sense of noticing events that occur on smaller time scales. High temporal acuity may be likened to higher temporal resolution. NMDA receptors may mediate perceptual information that is low temporal acuity, due to the fact that NMDA receptors take up to 100s of milliseconds to activate and have longer excitatory effects on the neuron. On the other hand, AMPA receptors may mediate experiences of higher temporal acuity, due to having rapid and short excitatory effects on the neuron, activating in the milliseconds or shorter range of temporal resolution. This hypothesis has implications for pathologies involving altered NMDAr and AMPAr function, such as schizophrenia, Alzheimer’s disease, psychedelics drug intoxication, and dissociative anesthetic intoxication. Besides this, the findings will provide reasoning for the development of novel biomarkers for NMDAr and AMPAr status in patients. The hypothesis is tested using 3 drugs, Theanine (NMDAr enhancer, AMPAr antagonist), Dextromethorphan (NMDAr antagonist), and Unifiram (AMPAr enhancer/AMPAkine). A flickering light will be presented to 4 experimental groups to observe whether AMPAkines allow users to observe usually unobservable light flickers.
This paper adventures into the realm of hallucinogenic drugs. Flicker resonance is explored as a possible mechanism for psychedelic fractals. Time dilation is touched on as well.
These ideas are an expansion of the project Flicker.
INTRODUCTION
NMDAr and AMPAr
Glutamate is a major excitatory neurotransmitter in the brain. There are two common glutamate receptor types that exist on very many of the neurons in the brain. They both work together in learning processes and modulation of these receptors has been found to alter perception. For example, NMDA receptor (NMDAr) antagonists produce intense perceptual changes and hallucinations (Krystal, D’Souza, Mathalon et al 2003). There are also anecdotal reports of AMPA receptor (AMPAr) agonists producing an increase in visual acuity and color sensation throughout many forums on the internet. One of the distinctions in the mechanisms of these two glutamate receptor types is in the speed in which they lead to neural excitation and excitatory decay. AMPAr produces a fast and short duration of excitatory effect, meanwhile NMDAr produces a slower and longer duration of excitatory effect, by many times more than AMPAr. The AMPAr produces short-lasting kinetics compared to NMDAr. While AMPAr remains open for a few milliseconds, NMDAr can stay open for hundreds of milliseconds and activates more slowly (Luscher & Malenka 2012). So the excitatory postsynaptic potentials (EPSP) produced by AMPAr will be far shorter than ones produced by NMDAr. While NMDAr-mediated EPSPs vary in duration, it has been argued that the longer EPSPs are critical for plasticity and Hebbian learning (Roberts & Romoa 1999). Hebbian learning is often described concisely as ‘neurons that fire together, wire together’. A longer duration of an active state neuron would create a wider window for which it may overlap with other active state neurons, in essence forming a co-incident event.
Found this example on google images, source is listed in the graphic. The image shows AMPAr EPSP on the left and NMDAr EPSP on the right.
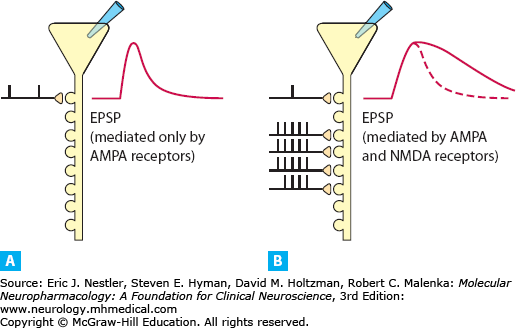
The NMDAr and AMPAr play a role in learning and memory (Tabone & Ramaswami 2012). NMDArs are blocked by magnesium ions, which prevent EPSPs from occurring. AMPAr activity produces effects that allow magnesium to be removed from the NMDAr, which then allows NMDAr to produce EPSPs (Luscher & Malenka 2012). NMDAr activation is required for long-term potentiation to occur, a mechanism that is critical in learning and long-term memory (Luscher & Malenka 2012). Blocking NMDAr impairs associative learning (Pallares, Nadal, Silvestre, & Ferre 1995) and memory (Farahmandfar, Akbarabadi, Bakhtazad, & Zarrindast 2017). NMDAr also appears to play a role in a process known as temporal summation (Wu, Ma, & Kelly 2004), where NMDAr antagonism can block temporal summation (Price, Mao, Frenk, & Mayer 1994). Temporal summation occurs when a neuron is stimulated repeatedly in a short time, where the EPSPs in some sense ‘stack’ and produce a summated effect (Felten, O’Banion, Maida, & Netter, 2016). Another kind of summation, spatial summation, involves two neurons stimulating a third neuron within a short time frame to produce a boosted or ‘summated’ EPSP on the third neuron. It isn’t clear if NMDAr is involved in spatial summation, but spatial summation seems to have temporal summation embedded in its’ concept. The third neuron is being stimulated within some time period and NMDAr may play the role of widening the time space that allows for response ‘stacking’. This reveals that NMDAr plays at least some role in temporal processing, perhaps helping to bind temporal events that don’t occur within the short AMPAr EPSP time scale.
Temporal Acuity
There is something known as the flicker fusion threshold, which is described as the frequency when a flickering light becomes too fast for one to notice the temporal gaps between each flicker. In essence, a flickering light that is above this threshold will appear as a solid light to humans. Having a higher threshold is thought to be associated with higher efficiency in non-color visual processing pathways (Brown et al 2018). Flicker fusion threshold may determine visual temporal acuity, which refers to the degree of temporal resolution one can be aware of. Those with higher temporal acuity will therefore perceive smaller units of time in comparison to those with low temporal acuity.
The difference in activation speed and duration observed between these two receptors could help to explain differences in temporal perception that occur in various circumstances such as drug intoxication and saccades. The difference in temporal visual perception will be termed temporal acuity, which refers specifically to the sensitivity to temporal events that occur in shorter distances of time. To have increased temporal acuity means to be able to detect perceptual changes that occur in smaller distances of time. It is thought that temporal acuity changes depending on the state of one’s mind. For example, during eye movements known as saccades, people are reportedly capable of observing the flickers of household lightbulbs, with frequencies as high as 2000Hz being observed between saccades (Wilkins 2014). This study also observed that 3000Hz was not observable by humans. Another study has found that a 500Hz flicker is observable during saccades (Binda, Cicchini, Burr, & Morrone 2009). These observations may be possible when summation ceases, so that time is no longer ‘blending’ or overlapping and one can observe flickering. This is explained further in the section titled temporal perception.
Saccades are particularly interesting because it is known that EEG frequencies track with saccadic movements. Saccadic movements reportedly produce as high as 600Hz EEG frequencies that last only for a brief moment during visual scanning (Brigo 2011). Saccades also have implications for schizophrenia, as patients with this disorder are reported to have altered saccadic movements (Avila, Weiler, Lahti et al 2002) and also hypofunctioning NMDArs (Coyle 2012). Ketamine, an NMDAr antagonist, has been shown to induce comparable saccadic changes as the ones observed in schizophrenics (Avila, Weiler, Lahti et al 2002). This suggests that NMDAr may be involved in saccadic eye movements. It may be that the fast frequencies observed during saccades are the result of the brain trying to update visual information as a novel scene is presented to the eyes during a shift of perspective.
EEG frequencies might have usefulness in checking AMPAr versus NMDAr activity dominance, where lower frequencies may occur during NMDAr dominant brain activity and higher frequencies may represent AMPAr dominant activity. Modulation of AMPAr and NMDAr has been observed to produce changes to EEG as well. AMPAr antagonists seem to increase slow waves and reduce faster frequencies (Routley, Singh, Hamandi, & Muthukumaraswamy 2017), presumably because AMPAr mediates faster frequencies. On the other hand, NMDAr antagonists reduce the power of slow frequencies and increase the power of faster frequencies which also correlated to subjective psychotic effects in humans (de la Salle, Choueiry, Shah et al 2016). This is likely due to NMDAr playing a role in slow waves. Another study showed that low magnesium facilitated slow wave oscillations, which was blocked by an NMDAr antagonist (Flint & Connors 1996). NMDAr agonists tend to produce seizures (Aldinio, French, & Schwarcz 1983), which complicates finding the role of NMDAr in slow wave activity, although there has been some research showing that an NMDAr agonist known as ibotenic acid can produce slow waves during sleep (Marini, Ceccarelli, & Mancia 2000). Another study found that NMDAr and AMPAr agonists both increased higher frequencies on the EEG (Fournier, Materi, Semba, & Rasmusson 2004). Finally, one study has found that NMDAr and AMPAr were both involved in generating slow waves while faster waves were almost exclusively AMPAr in an animal (barrel) cortex (Minlebaev, Ben-Ari, & Khazipov 2009). The ways in which these drugs impact EEG is likely complicated due to many downstream effects that would occur dose-dependently.
Cognition
Not only is schizophrenia relevant to this research, but so is Alzheimer’s disease (AD). In AD there have been treatments like NMDAr antagonists (Peskind, Potkin, Pomara et al 2006) and AMPAkines, which enhance AMPAr activity (Wezenberg, Verkes, Ruigt et al 2007). One of the more peculiar experimental therapies being studied for AD is stroboscopic entrainment therapy. There have been recent studies showing that giving stroboscopic therapy to AD mice with 40Hz strobe lights for 1 hour per day, for a week, produced a massive reduction (around 50%) in beta-amyloid plaques, which are abnormally built up in AD (Iaccarino, Singer, Martorell et al 2016). These plaques returned in the days following the therapy unfortunately. What’s more is that beta-amyloid itself binds to NMDAr as an agonist (Zhang, Li, Feng, & Wu 2016). This mechanism might also help to explain the loss of higher frequency (gamma oscillations – 20-50Hz) EEG readings that are observed in AD (Iaccarino, Singer, Martorell et al 2016). AMPAkines and strobe lights might force faster neuron activity, restoring the higher range of EEG activity and somehow reversing the build-up of beta-amyloid plaques. This also suggests that reduced AMPAr activity plays a role in the buildup of beta-amyloid. This makes sense as the early course of AD and aging are both shown to involve downregulation and impairment of AMPArs (Henley & Wilkinson 2013).
Dynorphins and Cognitive Impairment
This downregulation of AMPAr may be due to a neurochemical group known as dynorphins or just ‘dynorphin’. In mice, the removal of dynorphin genes prevents the age-related loss of specific AMPAr subunits, Glur1 and Glur2, in the hippocampus of rats (Ménard, Herzog, Schwarzer, & Quirion 2014). When AMPAr downregulates, this may somehow allow beta-amyloid to build up, although how this happens isn’t totally clear. It may be that AMPAr activity normally accelerates beta-amyloid removal by altering the NMDAr dynamics. This is speculation and an area that needs further research.
Dynorphin is also implicated in schizophrenia and Down’s syndrome as well. Schizophrenia shows elevated dynorphin (Heikkilä, Rimón, & Ternius, 1990) and accelerated brain aging (Hajek, Franke, Kolenic et al 2019). The same pattern is found in Down’s syndrome, with elevated dynorphin (Risser, You, Cairns et al 1996) and even elevated beta-amyloid (Lott, Head, Doran, & Busciglio 2006). Dynorphin has been associated to cognitive decline in multiple states: with aging (Ménard, Herzog, Schwarzer, & Quirion 2014), with alcohol use (Kuzmin, Chefer, Bazov et al 2013), with stress (Carey, Lyons, Shay et al 2009), and with a painful condition of the teeth (Yamada, Ono, Kubo et al 2013). The anti-cognitive effects of dynorphin are likely due to its ability to suppress plasticity (Wagner, Terman, & Chavkin 1993), block NMDAr (Chen, Gu & Huang 1995), and reduce glutamate release (Wagner, Terman, & Chavkin 1993).These patterns of dynorphin increasing, AMPAr function declining and rising beta-amyloid in these conditions involve learning and memory impairments suggest that there may be a convergent series of mechanisms implicated in general cognitive dysfunction and disorders involving cognitive dysfunction.
In the future, flicker tests could be used as biomarkers in relevance to cognitive function, much like we use markers like eye dilation. It could signify pharmacological mechanisms underlying cognitive dysfunction, which would suggest that the problem can be attenuated. There is some evidence of altered flicker fusion thresholds in patients with Alzheimer’s and vascular dementia (Curran et al 2014), although it isn’t much and more research would be needed.
Temporal Perception
Besides having implications for cognitive problems in schizophrenia, Alzheimer’s and Down’s syndrome, the flicker hypothesis has implications for the general mechanisms of temporal perception. Other mechanisms with known roles in temporal perception are D2 dopamine receptor and GABA (Meck 1996). D2 dopamine receptors were found to play a crucial role in time perception and also modulate glutamate release (Meck 1996). GABA also appears to play a role in time perception (Terhune, Russo, Near et al 2014).
Time sense might involve a specific mechanism or circuitry that is geared towards managing a subjective sense of how much time is passing, but it is also possible that time sense is measured by comparing how much awareness of perceptual or cognitive events occur between events with known temporal distances. For example, we may have trained ourselves to recognize how many observations or experiences can occur between the movements of a second-hand on an analog clock. If we begin to notice an increased number of experiences between second-hand movements, then we may feel that time is passing more slowly. We may have some kind of standardized and learned sense of time passing that correlates to how much we can observe, experience, or think between those events. An increase in the number of observations per second might appear to us as time expansion. If time sense is not specialized circuitry (or perhaps even if so) but instead is based on relative subjective noticing of the quantities of temporal events, then temporal acuity may mediate changes in time sense. The sense that time is slow could refer to increased visual information cycles being observed at the same rate. For example, AMPAr activity is capable of producing excitatory postsynaptic potential and decay 10x (probably even more) in the period of time that an NMDAr-induced neuron EPSP occurs. If the rate that EPSPs produce subjective experiences is constant, then time could seem 10x slower if AMPAr were dominant.
Psychedelic and dissociative hallucinogenic drugs are colloquially known to produce alterations to temporal perception, usually termed time dilation. This term refers to an expanded sense of time, which could potentially be explainable as heightened temporal acuity as described in the previous passage, although we should be cautious in assuming this. The subjective effects of psychedelic drugs are thought to occur by disinhibiting glutamate release via 5HT2a receptor and mGlur2 interactions (Moreno, Holloway, Albizu et al 2011). This means both AMPAr and NMDAr activity should increase. 5HT2a receptor agonism seems to both enhance (Dantsuji, Nakamura, Nakayama et al 2019) and partially inhibit NMDAr activity, functioning like a partial agonist of NMDAr (Arvanov, Liang, Russo, & Wang 1999). This might increase temporal summation, which I hypothesize may partly play a role in effects such as visual trailing (palinopsia), essentially the fusion element of flicker fusion. Decreased flicker fusion thresholds have been observed in people who used LSD (Abraham & Wolf 1988) and LSD is known to occasionally produce persisting palinopsia as well (Kawasaki & Purvin 1996). It could be that palinopsia and afterimages occur as effects of the prolonged EPSP of NMDArs, resulting in visual stimuli lingering in visual perception for longer than usual. In some sense, this may resemble a kind of ‘memory’ effect. This could be described as perceptual merging of temporal events, to observe the events that have occurred within a 100+ millisecond time span all simultaneously. Psychedelics also increase AMPAr activity indirectly through the release of glutamate, so it might be possible that psychedelics produce enhanced temporal acuity simultaneously to prolonged EPSPs. It may also be that these effects vary greatly with psychedelics, depending on dose or even fluctuating as the drug runs its’ course.
An artistic rendition of palinopsia.
NMDAr antagonists have been reported to induce time dilation and also a very peculiar effect known by users as ‘strobing’ or ‘flanging’ (Earth Erowid 2009). This effect is said to be like viewing the world through a stroboscope or viewing life as a stop-motion film. It is possible that some of this is due to becoming suddenly able to observe the flickering of household lights. It is possible that the drug reveals both the brain’s internal flickering and the flickering of external household lights. In this study, the question of whether users observe the flicker of lights or internal brain signaling could be answered if users’ response changes based on the frequency of the external lights. Normally, temporal summation and prolonged NMDAr EPSPs may prevent observations of both endogenous and exogenous flickering/strobing/flanging perceptions by filling in the gaps with something like palinopsia or motion blur. An Erowid writer suggests the strobing effect may be likened to akinetopsia, a condition where people lose their ability to perceive motion correctly (Earth Erowid 2009).
Flicker Resonance and Fractals
Serotonergic psychedelic drugs are said to produce fractal geometry with both open and closed eyes. The existence of the fractal effects of psychedelics may elude to endogenous brain flickering. It is possible that resonance from internal brain flickering generates such patterns. I’ll demonstrate this with a comparison of two videos.
Fractal resonance patterns from vibrations in liquid.

Consider that flickers are like waves, they have frequencies, they oscillate. People have found that the surface of water can form patterns that correspond to the frequency of the sound. The image above is known as cymatics, which is the study of sound and vibration made visible through some medium, like water or sand. It often involves using sound through water (or other mediums) so that the distortions of the water reveal the shape of the sound waves.
You can observe this in the video below.
Starting at 20 seconds and above, the patterns become increasingly chaotic and fractal-like.
It is possible to induce similar patterns observed in this video above while sober, both closed and open-eyed visuals using a strobe light. A strobe light has a frequency, much like this sound, and so the fractals induced by the strobe light may be resonance patterns like observed on the surface of the water. In this case, the medium is not water but your own perception. The internal brain flicker may produce resonating patterns by interfering with other neurons that flicker at differing frequencies. Flicker resonance may ultimately produce the kind of fractals people observe on psychedelic drugs.
One may wonder if perception is a kind of virtual liquid medium.
There is a video below that you can test on yourself. Be careful, as this video can induce seizures in those susceptible. The frequency observed in this strobe light simulation is 12Hz. Compare the geometry that you observe at the 12 second mark in the first video, as they seem very similar, honeycomb like shapes. It isn’t clear what frequency is occurring in the liquid medium at the 12 second mark, but it may potentially be near 12 Hz.
!!SEIZURE WARNING!! Here is the video link.
I did not embed the above video because I do not want anyone to accidentally click play and experience adverse effects, such as a seizure. Click at your own risk!
The fractals that appear on these drugs could be fractals that normally exist within the brain but don’t make it to perception. These patterns may be so repetitive at normal brain frequencies that the brain learns to suppress the hyper-predictable stimuli due to the irrelevance and constancy of the effect. The psychedelic drug may then reverse the conditioned state of perception, as if one has not yet learned to suppress this information, like an infant. It is possible that uncommon frequencies of brain processing unveil these patterns simply due to being uncommon frequencies, ones that haven’t yet been able to ‘train out’ of perception. Many report enhanced sensory acuity on psychedelic drugs, which may suggest this reduction of filtering is partly at play in the development of visual fractals.
Alternatively, it may be that certain higher frequencies are required for the observation of visual geometry sober and on psychedelics. It could be that regions involved in perception have de-synchronized activity with other visual processing regions in such a way that produces resonance patterns. It is not entirely clear why fractal geometry occurs with psychedelic drugs.
Lastly, you can observe similar resonance patterns in the top video and also observe a slow-motion of cymatics in the lower video.
METHODS
The focus of this experiment is to test the hypothesis that AMPA receptors mediate the perception of highly acute temporal events. In this experiment we will use 3 different lightbulbs that ‘flicker’ at 3 different speeds, slow, medium, and fast. I specifically hypothesize that low NMDA receptor activity in combination with high AMPA receptor activity will allow people to observe high frequency light flickers, demonstrating a role of these mechanisms in temporal acuity. Three drugs and one drug combination will be tested. The first will be dextromethorphan (DXM), an NMDAr antagonist. This drug is expected to enhance the perceptibility of high frequency flickering, thus enhancing temporal acuity. The second will be Theanine, an NMDAr enhancer and AMPAr inhibitor. This drug is expected to decrease or not impact temporal acuity. The third drug will be an AMPAr agonist known as unifiram. The expectation is that this drug will enhance perception of high frequency light flickers and thus temporal acuity. Lastly, a combination of the AMPAr agonist and NMDAr antagonist will be tested, to test whether inhibition of NMDAr is required for perception of temporally acute events. The expectation is that out of all other drug conditions, this combination will produce the strongest enhancement of temporal acuity perception.
Participants
The participants will be chosen among healthy university students and equal age Facebook users who have no mental health diagnoses, in order to avoid confounds since we already observe changes to temporal perception in schizophrenia. Those with epilepsy will be excluded as well, because high NMDAr activity has been implicated in seizures (Dingledine, McBain, & McNamara 1990) and the flicker light conditions might be a seizure risk. It seems unlikely that university students have any particular difference in temporal acuity mechanisms, so the university students should suffice for this study. The students will be discovered through advertisements outside of classrooms and also through Facebook targeted advertisements that are local as to minimize travel distance for participants. The minimum age will be 24 years old.
The participants will be randomized to the drug condition so that they do not choose which condition, to reduce bias.
Light Conditions
The light is LED based and has a controlled flicker rate that can be modulated at 10Hz sensitivity. The light machine is connected to a computer that uses a random number generator to determine which flicker rate is presented to the participant. The frequencies include 10Hz and every next 10Hz until 100Hz. So for example, 10, 20, 30, and so on until 100 Hz. Beyond 100 Hz the frequencies will be set to 100 Hz intervals, for example, 100, 200, 300, 400, 500. . . up to 3000 Hz. The intervals of 10Hz for lower frequencies is important because these translate to the frequency ranges of EEG measurements. Going beyond 100Hz will likely produce less observed flickers in the participants. It is thought that frequencies as high as 2000Hz are observable by humans, but 3000Hz produced no observable flicker, so the frequency range used in this study stops at 3000Hz. The presumed undetectable 3000Hz frequency can be used to test if participants are potentially observing flickers that aren’t due to the light condition but instead due to their own perception flickering. It is not guaranteed that 3000Hz is entirely unobservable though, so we must remain cautious here. The doses used for the drug conditions are not intended to produce strobing effects on general vision, although those doses may be necessary if no effect is observed. To disentangle the interaction of temporal acuity and NMDAr/AMPAr may prove difficult if some participants report detecting all light conditions.
Drug Conditions
The use of AMPAr enhancing drugs poses an issue, due to the way AMPAr activity might accelerate magnesium removal from the NMDAr receptor. AMPAr enhancing drugs may then produce increases in NMDAr activity, which has been observed with AMPAkine drugs (Knafo & Esteban, 2015). Because of this, the combination of AMPAr enhancing and NMDAr inhibiting drugs might be better suited to revealing how AMPAr activity impacts temporal acuity. The experiment will still test a group with the isolated AMPAkine drug, Unifiram, in order to observe whether or not an effect is produced. The dose used for Unifiram will be 10mg, which should produce very noticeable effects in most users.
Another drug condition will involve Theanine, which is known to enhance NMDAr activity, seemingly as a partial agonist (Wakabayashi, Numakawa, Ninomiya 2012), and also function as an AMPAr antagonist (Kakuda, Nozawa, Sugimoto, & NIINO 2002). This could help to isolate the effects of NMDAr without having AMPAr activity as a confound variable. There are still other mechanisms of Theanine that might introduce confounds, such as alterations to monoamines (Shen, Yang, Wu et al 2019), but this may occur through glutamatergic modulating mechanisms and thus not entirely pose an issue. The dose used for the Theanine condition will be 200mg, which is a common dose used in supplements and decently strong, which should produce sufficient effects on NMDAr and AMPAr activity.
Another drug condition is dextromethorphan (DXM) which has been commonly used to research NMDAr blockade in the literature. This drug is favorable because it is widely available as an over the counter medication in most places and is also legal. This makes the drug accessible. One issue is that DXM has other known effects, such as modulation of the serotonin system which may introduce confounds (McFee, Mofenson, & Caraccio 2000). The dose used for the DXM condition will be 280mg, which should not produce strong strobing effects while also being sufficient enough to produce noticeable effects on perception.
Lastly, a condition involving both DXM and Unifiram will be given, to produce a state in which AMPAr activity is enhanced while NMDAr activity is suppressed. This will help reduce the problem of AMPAr stimulation enhancing NMDAr activity so that it can be determined whether or not AMPAr itself is responsible for any observed changes. Unifiram will be given 45 minutes after DXM in order to ensure that both doses become active at around the 1.5 hour mark, where the testing will begin. The doses used for this condition will be 280mg DXM and 10mg Unifiram.
There will also be a placebo group who is given a sugar pill and engages the experiment at the 45 minute post-dose mark.
Procedure
The participants will be scheduled to visit the laboratory at a designated time. The participants will be instructed to be fasted for at least 3-4 hours before arriving to the laboratory. Before the drug is given, participants will told of possible side effects and risks of the drug they may have gotten. A medical team will be on-hand to administer a benzodiazepine (lorazepam) in case of adverse reactions such as anxiety, panic, psychosis, or seizures. They will also be fitted with an EEG, so that we can observe changes that the drug condition brings on. This will also be measured continuously throughout the test phase as well.
The drug conditions will be administered to each group of participants at the beginning. The participants will be shown an example of obvious flickering and how to operate the button machine. After the instructions are made clear, the participants will undergo a trial version in which the flickering is obvious, to ensure that the participants fully understand the rules. If this is successful, the participants will be directed towards a totally darkened room containing the light machine. This will occur 1 hour after the dose for conditions involving Theanine and Unifiram, to ensure that the drug has begun taking effects. The DXM group will start at 1.5 hours due to an expected delay in the onset of effects, something that is commonly reported. The DXM/Unifiram group will start at 1.5 hours as well, with Unifiram being dosed 45 minutes after DXM, in order to ensure both drugs are active during the testing phase.
The participants will enter a darkened room with one of the lab assistants. During the experiment, the participants will press a green button if they detect flickering of the lights. If the light appears continuous and solid, then they are instructed to press a red button to indicate that the light does not appear to be flickering. The frequency of the light flicker will change every 5 seconds and be followed by 1 second of darkness between each light test. The order of the flicker frequency tests will be randomized using computer software.
Analysis
The hypothesized results are as follows:
- The Unifiram condition may enhance temporal acuity, even with enhancement of NMDAr. This will result in participants observing higher frequency flickers than the other experimental groups. This is expected to increase fast wave EEG, in the gamma range.
- The Theanine condition may decrease temporal acuity, due to enhancing NMDAr activity and reducing AMPAr activity. This condition may enhance temporal summation and produce ‘perceptual merging’ of flickers, so that the light appears as a continuous stream. This is expected to reduce fast wave and increase slow waves as measured by the EEG.
- The DXM condition may result in enhanced temporal acuity, due to indirectly enhancing AMPAr by inducing glutamate release and also reduce temporal summation or ‘perceptual merging’ of flickers by reducing NMDAr activity. This is expected to increase fast wave EEG measurements and decrease slow waves.
- The DXM/Unifiram condition may increase temporal acuity, even more so than the other experimental groups. This is expected because NMDAr activity will be reduced while AMPAr activity is increased, which should promote mostly faster signaling of neurons. This is expected to produce a stronger effect on fast wave measurements on the EEG than other experimental groups.
If these hypotheses are supported by the data, this means we could begin to push forward with new forms of biomarkers in the study of cognition. For example, we can begin to test whether various mental health conditions involve changes in temporal acuity, starting with disorders known to involve alterations to NMDAr and AMPAr function. This could potentially help further detect disorders of NMDAr and AMPAr activity. Some researchers have already noted that flicker fusion threshold may be a useful indicator for traumatic brain injury accompanied by light sensitivity, a specific type of dyslexia, and Alzheimer’s/dementia (Chang, Ciuffreda, & Kapoor 2007; Peters et al 2020; Curran et al 2014).
If the hypotheses are wrong, then there may alternative explanations for effects observed. For example, it could be that the strobing effects observed on NMDAr antagonists are the result of AMPAr desensitizing due to the extra burden on AMPAr from a lack of NMDAr activity. As AMPAr desensitizes, it may produce gaps in neural activity that are observable by other regions of the brain that interact with the neural gapping. This outcome is not necessarily contradictory to the hypotheses though, but it would suggest that the flicker isn’t necessarily representative of increased temporal acuity.
Another possibility is that increased temporal acuity does not occur evenly throughout the brain, resulting in some regions having enhanced temporal acuity and the ability to observe the lack of acuity in other regions that interact with the high acuity region. This would be like observing a low frame-rate video with your high frame rate vision. The gaps between frames become increasingly apparent the lower the video frame rate is. If DXM groups report that all lights are flickering, this could be a serious problem because it isn’t possible to conclude whether this is due to the person actually observing the flicker or if it is an observation of internal neurological flickering. Future research can begin to associate temporal acuity to various cognitive states, such as mania, depression, ADHD, psychosis, Alzheimer’s, and so on.
. . .
Special thanks to the six patrons: Melissa Bradley, Morgan Catha, Niklas Kokkola, Abhishaike Mahajan, Riley Fitzpatrick, and Charles Wright! Abhi is also the artist who created the cover image for Most Relevant. Please support him on instagram, he is an amazing artist! I’d also like to thank Annie Vu, Chris Byrd, and Kettner Griswold for your kindness and making these projects and the podcast possible through your donations.
If you liked this, follow me on
If you’d like to support these projects and help fund potential research like this, check out this page.
You can also follow the discussion for this post on Reddit:

CITATIONS
- Abraham, H. D., & Wolf, E. (1988). Visual function in past users of LSD: Psychophysical findings. Journal of Abnormal Psychology, 97(4), 443.
- Aldinio, C., French, E. D., & Schwarcz, R. (1983). The effects of intrahippocampal ibotenic acid and their blockade by (−) 2-amino-7-phosphonoheptanoic acid: morphological and electroencephalographical analysis. Experimental brain research, 51(1), 36-44.
- Arvanov, V. L., Liang, X., Russo, A., & Wang, R. Y. (1999). LSD and DOB: interaction with 5‐HT2A receptors to inhibit NMDA receptor‐mediated transmission in the rat prefrontal cortex. European Journal of Neuroscience, 11(9), 3064-3072.
- Avila, M. T., Weiler, M. A., Lahti, A. C., Tamminga, C. A., & Thaker, G. K. (2002). Effects of ketamine on leading saccades during smooth-pursuit eye movements may implicate cerebellar dysfunction in schizophrenia. American Journal of Psychiatry, 159(9), 1490-1496.
- Binda, P., Cicchini, G. M., Burr, D. C., & Morrone, M. C. (2009). Spatiotemporal distortions of visual perception at the time of saccades. Journal of Neuroscience, 29(42), 13147-13157.
- Brigo, F. (2011). Lambda waves. American journal of electroneurodiagnostic technology, 51(2), 105-113.
- Brown, A., Corner, M., Crewther, D. P., & Crewther, S. G. (2018). Human flicker fusion correlates with physiological measures of magnocellular neural efficiency. Frontiers in human neuroscience, 12, 176.
- Carey, A. N., Lyons, A. M., Shay, C. F., Dunton, O., & McLaughlin, J. P. (2009). Endogenous κ opioid activation mediates stress-induced deficits in learning and memory. Journal of Neuroscience, 29(13), 4293-4300.
- Chang, T. T. L., Ciuffreda, K. J., & Kapoor, N. (2007). Critical flicker frequency and related symptoms in mild traumatic brain injury. Brain injury, 21(10), 1055-1062.
- Chen, L., Gu, Y., & Huang, L. Y. (1995). The mechanism of action for the block of NMDA receptor channels by the opioid peptide dynorphin. Journal of Neuroscience, 15(6), 4602-4611.
- Coyle, J. T. (2012). NMDA receptor and schizophrenia: a brief history. Schizophrenia bulletin, 38(5), 920-926.
- Curran, S., Wilson, S., Musa, S., & Wattis, J. (2004). Critical Flicker Fusion Threshold in patients with Alzheimer’s disease and vascular dementia. International journal of geriatric psychiatry, 19(6), 575-581.
- Dantsuji, M., Nakamura, S., Nakayama, K., Mochizuki, A., Park, S. K., Bae, Y. C., … & Inoue, T. (2019). 5‐HT2A receptor activation enhances NMDA receptor‐mediated glutamate responses through Src kinase in the dendrites of rat jaw‐closing motoneurons. The Journal of physiology, 597(9), 2565-2589.
- de la Salle, S., Choueiry, J., Shah, D., Bowers, H., McIntosh, J., Ilivitsky, V., & Knott, V. (2016). Effects of ketamine on resting-state EEG activity and their relationship to perceptual/dissociative symptoms in healthy humans. Frontiers in pharmacology, 7, 348.
- Dingledine, R., McBain, C. J., & McNamara, J. O. (1990). Excitatory amino acid receptors in epilepsy. Trends in pharmacological sciences, 11(8), 334-338.
- Earth Erowid (2009). The DXM Stop Motion Effect (‘Strobing’ or ‘Flanging’) Terminology for Interference with the Visual Perception of Motion. Retrieved June 27, 2020, from https://erowid.org/chemicals/dxm/dxm_effects1.shtml
- Farahmandfar, M., Akbarabadi, A., Bakhtazad, A., & Zarrindast, M. R. (2017). Recovery from ketamine-induced amnesia by blockade of GABA-A receptor in the medial prefrontal cortex of mice. Neuroscience, 344, 48-55.
- Felten, D. L., O’Banion, M. K., Maida, M. S., & Netter, F. H. (2016). Neurons and Their Properties. In Netter’s Atlas of Neuroscience (pp. 1-42). Philadelphia, PA: Elsevier.
- Flint, A. C., & Connors, B. W. (1996). Two types of network oscillations in neocortex mediated by distinct glutamate receptor subtypes and neuronal populations. Journal of Neurophysiology, 75(2), 951-957.
- Fournier, G. N., Materi, L. M., Semba, K., & Rasmusson, D. D. (2004). Cortical acetylcholine release and electroencephalogram activation evoked by ionotropic glutamate receptor agonists in the rat basal forebrain. Neuroscience, 123(3), 785-792.
- Hajek, T., Franke, K., Kolenic, M., Capkova, J., Matejka, M., Propper, L., … & Kopecek, M. (2019). Brain age in early stages of bipolar disorders or schizophrenia. Schizophrenia bulletin, 45(1), 190-198.
- Heikkilä, L., Rimón, R., & Ternius, L. (1990). Dynorphin A and substance P in the cerebrospinal fluid of schizophrenic patients. Psychiatry research, 34(3), 229-236.
- Henley, J. M., & Wilkinson, K. A. (2013). AMPA receptor trafficking and the mechanisms underlying synaptic plasticity and cognitive aging. Dialogues in clinical neuroscience, 15(1), 11.
- Iaccarino, H. F., Singer, A. C., Martorell, A. J., Rudenko, A., Gao, F., Gillingham, T. Z., … & Adaikkan, C. (2016). Gamma frequency entrainment attenuates amyloid load and modifies microglia. Nature, 540(7632), 230-235.
- Kakuda, T., Nozawa, A., Sugimoto, A., & NIINO, H. (2002). Inhibition by theanine of binding of [3H] AMPA,[3H] kainate, and [3H] MDL 105,519 to glutamate receptors. Bioscience, biotechnology, and biochemistry, 66(12), 2683-2686.
- Kawasaki, A., & Purvin, V. (1996). Persistent palinopsia following ingestion of lysergic acid diethylamide (LSD). Archives of Ophthalmology, 114(1), 47-50.
- Knafo, S., & Esteban, J. A. (2015). Molecular Mechanisms of Drug-Induced Cognitive Enhancement. In Cognitive Enhancement (pp. 43-59). Academic Press.
- Krystal, J. H., D’Souza, D. C., Mathalon, D., Perry, E., Belger, A., & Hoffman, R. (2003). NMDA receptor antagonist effects, cortical glutamatergic function, and schizophrenia: toward a paradigm shift in medication development. Psychopharmacology, 169(3-4), 215-233.
- Kuzmin, A., Chefer, V., Bazov, I., Meis, J., Ögren, S. O., Shippenberg, T., & Bakalkin, G. (2013). Upregulated dynorphin opioid peptides mediate alcohol-induced learning and memory impairment. Translational psychiatry, 3(10), e310-e310.
- Lott, I. T., Head, E., Doran, E., & Busciglio, J. (2006). Beta-amyloid, oxidative stress and down syndrome. Current Alzheimer Research, 3(5), 521-528.
- Lüscher, C., & Malenka, R. C. (2012). NMDA receptor-dependent long-term potentiation and long-term depression (LTP/LTD). Cold Spring Harbor perspectives in biology, 4(6), a005710.
- Marini, G., Ceccarelli, P., & Mancia, M. (2000). Effects of bilateral microinjections of ibotenic acid in the thalamic reticular nucleus on delta oscillations and sleep in freely‐moving rats. Journal of sleep research, 9(4), 359-366.
- McFee, R. B., Mofenson, H. C., & Caraccio, T. R. (2000). Dextromethorphan: another” ecstasy”?. Archives of Family Medicine, 9(2), 123.
- Meck, W. H. (1996). Neuropharmacology of timing and time perception. Cognitive brain research, 3(3-4), 227-242.
- Ménard, C., Herzog, H., Schwarzer, C., & Quirion, R. (2014). Possible role of dynorphins in Alzheimer’s disease and age-related cognitive deficits. Neurodegenerative Diseases, 13(2-3), 82-85.
- Minlebaev, M., Ben-Ari, Y., & Khazipov, R. (2009). NMDA receptors pattern early activity in the developing barrel cortex in vivo. Cerebral Cortex, 19(3), 688-696.
- Moreno, J. L., Holloway, T., Albizu, L., Sealfon, S. C., & González-Maeso, J. (2011). Metabotropic glutamate mGlu2 receptor is necessary for the pharmacological and behavioral effects induced by hallucinogenic 5-HT2A receptor agonists. Neuroscience letters, 493(3), 76-79.
- Pallares, M. A., Nadal, R. A., Silvestre, J. S., & Ferre, N. S. (1995). Effects of ketamine, a noncompetitive NMDA antagonist, on the acquisition of the lever-press response in rats. Physiology & behavior, 57(2), 389-392.
- Peskind, E. R., Potkin, S. G., Pomara, N., Ott, B. R., Graham, S. M., Olin, J. T., … & Memantine MEM-MD-10 Study Group. (2006). Memantine treatment in mild to moderate Alzheimer disease: a 24-week randomized, controlled trial. The American Journal of Geriatric Psychiatry, 14(8), 704-715.
- Peters, J. L., Bavin, E. L., Brown, A., Crewther, D. P., & Crewther, S. G. (2020). Flicker fusion thresholds as a clinical identifier of a magnocellular-deficit dyslexic subgroup. Scientific reports, 10(1), 1-10.
- Price, D. D., Mao, J., Frenk, H., & Mayer, D. J. (1994). The N-methyl-D-aspartate receptor antagonist dextromethorphan selectively reduces temporal summation of second pain in man. Pain, 59(2), 165-174.
- Religa, D., Laudon, H., Styczynska, M., Winblad, B., Näslund, J., & Haroutunian, V. (2003). Amyloid β pathology in Alzheimer’s disease and schizophrenia. American Journal of Psychiatry, 160(5), 867-872.
- Risser, D., You, Z. B., Cairns, N., Herrera-Marschitz, M., Seidl, R., Schneider, C., … & Lubec, G. (1996). Endogenous opioids in frontal cortex of patients with Down syndrome. Neuroscience letters, 203(2), 111-114.
- Roberts, E. B., & Ramoa, A. S. (1999). Enhanced NR2A subunit expression and decreased NMDA receptor decay time at the onset of ocular dominance plasticity in the ferret. Journal of Neurophysiology, 81(5), 2587-2591.
- Routley, B. C., Singh, K. D., Hamandi, K., & Muthukumaraswamy, S. D. (2017). The effects of AMPA receptor blockade on resting magnetoencephalography recordings. Journal of Psychopharmacology, 31(12), 1527-1536.
- Shen, M., Yang, Y., Wu, Y., Zhang, B., Wu, H., Wang, L., … & Chen, J. (2019). L‐theanine ameliorate depressive‐like behavior in a chronic unpredictable mild stress rat model via modulating the monoamine levels in limbic–cortical–striatal–pallidal–thalamic‐circuit related brain regions. Phytotherapy Research, 33(2), 412-421.
- Tabone, C. J., & Ramaswami, M. (2012). Is NMDA receptor-coincidence detection required for learning and memory?. Neuron, 74(5), 767-769.
- Terhune, D. B., Russo, S., Near, J., Stagg, C. J., & Kadosh, R. C. (2014). GABA predicts time perception. Journal of Neuroscience, 34(12), 4364-4370.
- Wagner, J. J., Terman, G. W., & Chavkin, C. (1993). Endogenous dynorphins inhibit excitatory neurotransmission and block LTP induction in the hippocampus. Nature, 363(6428), 451-454.
- Wakabayashi, C., Numakawa, T., Ninomiya, M., Chiba, S., & Kunugi, H. (2012). Behavioral and molecular evidence for psychotropic effects in L-theanine. Psychopharmacology, 219(4), 1099-1109.
- Wezenberg, E., Verkes, R. J., Ruigt, G. S., Hulstijn, W., & Sabbe, B. G. (2007). Acute effects of the ampakine farampator on memory and information processing in healthy elderly volunteers. Neuropsychopharmacology, 32(6), 1272-1283.
- Wilkins, A. (2014). Opinion: CFF is a seductive but misleading concept. Lighting Research and Technology, 46(4), 368.
- Wu, S. H., Ma, C. L., & Kelly, J. B. (2004). Contribution of AMPA, NMDA, and GABAA receptors to temporal pattern of postsynaptic responses in the inferior colliculus of the rat. Journal of Neuroscience, 24(19), 4625-4634.
- Yamada, K., Ono, Y., Kubo, K. Y., Yamamoto, T., & Onozuka, M. (2013). Occlusal disharmony transiently impairs learning and memory in the mouse by increasing dynorphin A levels in the amygdala. The Tohoku Journal of Experimental Medicine, 230(1), 49-57.
- Zhang, Y., Li, P., Feng, J., & Wu, M. (2016). Dysfunction of NMDA receptors in Alzheimer’s disease. Neurological Sciences, 37(7), 1039-1047.