Some of the mechanisms behind darkness adaptation overlap with illusory motion. These mechanisms may be tied to the mechanisms of psychedelic drugs. It may be that psychedelics amplify an endogenous night vision system, which causes certain visual distortions like illusory motion. Here is the evidence behind this possibility.
Most of you have probably heard that there are rods and cones in the eye. The rods deal with light levels, also known as luminance (which is basically brightness levels). The rods do not deal with color perception. Color perception is driven by the cones in the eye. The connection between psychedelics and illusory motion focuses on the mechanisms underlying rod-based perception.
The rods and cones send signals that travel through distinct visual systems, namely the dorsal and ventral streams respectively. The dorsal stream moves up the visual cortex and into the parietal lobe. This stream deals with “where” information like depth, location, and movement. This stream is non-colored. The other system, known as the ventral stream, moves from the visual cortex towards the temporal lobe. This stream deals with “what” information, such as object identification. This stream is colored.
Since we are focusing on the rods, we will focus on the dorsal stream which deals with location and motion perception. Location and motion are key here since psychedelic visuals involve modifications to both of these. People on psychedelics experience illusory motion. To see an example of an image that induces sober illusory motion, just look below! This is the rotating snake illusion.

Something you can observe about this illusion is that it mostly occurs when you pan your vision across it. If you try to hold your eyes still, the image seems to cease its’ illusory motion. Another key observation is that this illusory motion occurs predominantly in peripheral vision.
What can cause this? One explanation is that decreased spatial resolution of information sent to the “where” or dorsal visual stream results in problems determining where the stimuli is exactly, and as you move your eyes around, the predicted locations change, which tells your brain that the stimuli has moved locations.
This decreased spatial resolution is due to rod convergence. The rods in the eye converge, meaning multiple rods may feed into fewer bipolar cells and eventually towards the dorsal visual stream in the brain (Purves et al. 2001). You can see an example of rod convergence below. While cones mostly have a one-to-one link to bipolar and ganglion cells, the rods connect in groups to single bipolar cells.
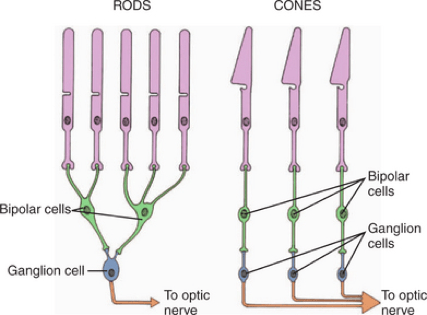
This convergence results in lower spatial resolution but higher sensitivity to light (Purves et al. 2001). The lower spatial resolution is due to the many spatially-placed rods only fueling a single bipolar or ganglion cell. The increased sensitivity to light is a result of the rods combining and summating their response to light in order to drive a single bipolar cell (Purves et al. 2001). This means that the threshold needed for the stimulation of the bipolar cell can be reached far more easily because multiple rods can stack together. Peripheral vision involves more rod convergence (Masland 2017) and has a lower density of rods, meaning that spatial resolution is further decreased (Gloriani & Schütz 2019).
Most importantly to this puzzle, Wikipedia says (don’t worry, academic sources to support this are provided further on) low light increases rod convergence to boost light sensitivity in the darkness:
In sufficiently bright light, convergence is low, but during dark adaptation, convergence of rod signals boost. This is not due to structural changes, but by a possible shutdown of inhibition that stops convergence of messages in bright light. If only one eye is open, the closed eye must adapt separately upon reopening to match the already adapted eye.
We will come back to this soon.
Remember, the illusion occurs mostly when we move our eyes. Since spatial resolution is low due to rod convergence in peripheral vision, spatial location must be predicted since it cannot be accurately observed by the eye. While holding still, a stable unchanging prediction is formed in our perception, but moving our eyes causes us to update the predicted location of the peripheral vision imagery. The change of location is assumed to be movement by the brain. So as we move our eyes across the image, the location of certain stimuli appear to keep changing places, and our brain registers it as the stimuli moving around.
Psychedelics are known to produce illusory motion similar to the rotating snakes illusion. At EffectIndex they have termed this effect “drifting”. An example is shown below:
Since psychedelics are known to dilate our eyes, something that occurs in low light conditions, I wondered if these drugs are stimulating a darkness adaptation system in the brain and retina. These drugs may tell our brain that we are in low light conditions and that we need to take measures to increase light sensitivity by dilating our eyes to allow more light in and also to increase convergence to amplify light sensitivity.
There is some evidence that psychedelics and serotonin may play a role in vision. Psychedelics work by stimulating 5HT2a and other serotonin receptors (Inserra, De Gregorio, & Gobbi 2021). There seems to be a role for serotonin and 5HT2a receptors in the retina of several species (Masson 2019), indicating a potential role in vision at the early retinal stages. It seems that the role of serotonin in the retina may even be rod-specific. From a paper about the role of serotonin in the retina (Jin & Brunken1998):
These data suggest that the serotoninergic system in retina is (1) specifically associated with rod-related pathways; and (2) exerts a powerful modulatory force over information transfer in the retina. Together these observations suggests that serotonin plays an important physiological role in modulating retinal processing.
. . .
Thus, it is tempting to speculate that serotonin may act as a dark-adapting
neurotransmitter, enhancing transmission through the rod pathway
either directly by modifying the output of rod bipolar cells or
indirectly by opposing the release or the effects of dopamine on the
retinal network.
Serotonin appears to be prominently involved in amacrine cell activity of the eye (Masson 2019). Amacrine cells are involved in lateral processing in the eye. You can see this in the image linked below. Amacrine cells seem to synthesize and release serotonin (Masson 2019). This is notable because amacrine cells play a role in modulating convergence and dark adapted vision (Demb & Singer 2012).
Amacrine cells can be observed in magenta. Notice that they are stretched out perpendicular to the majority of other cell types.
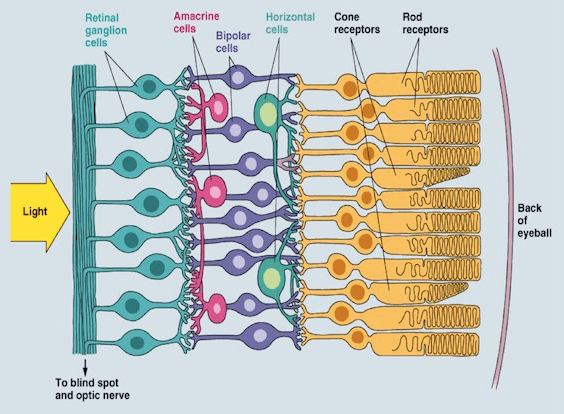
A study has explored the role of amacrine cell in dark adaptation (Demb & Singer 2012):
AIIs (amacrine cell subtype) themselves are remarkably sensitive to light: in the dark-adapted retina, AIIs respond reliably to dim flashes of light sufficient to isomerize single rhodopsin molecules in fewer than 1 in 3 to 1 in 100 rods (varying by species) (Pang et al., 2004; Trexler et al., 2005; Dunn et al., 2006). This sensitivity arises in part from the convergence of hundreds of rod outputs onto a single AII. Furthermore, electrical coupling between AIIs enhances the effective convergence of rods onto AIIs by permitting AIIs to be depolarized by rod bipolars that are not directly presynaptic (Sterling et al., 1988; Bloomfield et al., 1997; Pang et al., 2004).
The reduced coupling between AIIs (amacrine cell type) at photopic (daylight) intensities is thought to limit the lateral spread of neural signals in the inner retina, thereby improving the spatial acuity of bipolar, amacrine and ganglion cell receptive fields.
This suggests that specific types of amacrine cells may function to modulate how much convergence occurs depending on light levels. This is particularly interesting because of the proposed role serotonin has in amacrine cells and dark adaptation.
One study mentioned that serotonin may enhance dark adaptation by enhancing dendritic arborization which may increase visual sensitivity (Fosser, Brusco, & Ríos, 2005). This enhanced dendritic arborization may enhance convergence by increasing the number of cell branches (dendrites) that synapse rods, amacrine cells, and bipolar cells together. It may even specifically enhance amacrine cell coupling. Serotonergic psychedelics enhance dendritic arborization as well (Ly et al. 2018). Which might suggest that psychedelics enhance dendritic arborization in the retina, producing enhanced convergence and an increase in illusory motion. The study on serotonin and dark adaptation here seems to be over the course of consistent darkness and this may reflect a longer term plastic effect, such as increasing one’s capacity for visual sensitivity at low light, as opposed to rapid dendritic arborization.
Visual Snow
Perhaps the visual snow that some psychedelic users are left with after consuming the drug is a byproduct of alterations to dark and light adaptation. Visual snow seems somewhat normal under low light conditions. Th visual snow after drug use could be due to developing tolerance to night vision mechanisms, causing one to have impaired vision in darkness from reduced light sensitivity. For others, they might have residual effects that involve lasting darkness adaptation hypersensitivity, like illusory motion and glowy light-sensitive perception.
In support of this, it seems that filtering noise might be another role that amacrine cells play in the retina. Looking again at the study by Demb and Singer:
Though the convergence within the rod bipolar pathway in part underlies its sensitivity, convergence also increases the noise of neural responses by the square root of the number of inputs (Baylor et al., 1984; Sterling et al., 1988; Smith & Vardi, 1995). Gap junctions, by acting as low-pass filters, can average uncorrelated noise and improve the signal-to-noise ratio (S/N) in instances in which coupled cells receive correlated input (Lamb & Simon, 1976; Tessier-Lavigne & Attwell, 1988). Thus, it is thought that the AII network serves a noise-reducing role in rod-mediated vision.
This might also explain why vision under psychedelics can appear so clean and pristine. There is a sense of visual clarity that is often reported by users. It may be noise-reduction mechanisms. Then after the drug has faded, noise may enter the system much more. It would be interesting to see if anticholinergics increase noise and if this noise plays a role in misperceptions and hallucinations.
As an anecdote of dark adaptation and convergence illusions, I’ve noticed that if I stay in a dark room for 30 minutes and then go into the restroom with the lights on, the walls kind of shift and things look distorted for a while, very much like the effects of psychedelics. This could be due to darkness adaptations involving enhanced convergence of rods. This illusory motion only seems to last a minute or two at most. Go try this out for yourself and report back!
Another implication is with migraines, which involve light sensitivity and aura effects. Though, I suspect these may not necessarily share the same mechanism. I’ve argued that seizures, migraines, and psychedelics all share certain mechanisms that might be at play in the auras and visual effects of these conditions. You can read about that in 2-D Vision After Noopept.
The effects of psychedelics on vision are likely not only due to convergence alone. People report colors being brighter and many other effects that wouldn’t be explained by convergence, though could be luminance amplification. It is also possible that the motion perception illusions occurring on psychedelics involve multiple layers of the perceptual process and not just the retina as well.
. . .
If you found this enjoyable, consider joining the Patreon! I’ve been posting detailed experience reports with my adventures using prescription ketamine. Also. someone sent me an EEG device to collect data on ketamine-induced brainwave changes which I’ve started posting there too. I also post secret mini podcasts. You can find the publicly available podcasts here by the way!
Special thanks to the 12 patrons: Idan Solon, David Chang, Jack Wang, Richard Kemp, Milan Griffes, Alex W, Sarah Gehrke, Melissa Bradley, Morgan Catha, Niklas Kokkola, Riley Fitzpatrick, and Charles Wright! Abhi is also the artist who created the cover image for Most Relevant. Please support him on instagram, he is an amazing artist! I’d also like to thank Alexey Guzey, Annie Vu, Chris Byrd, and Kettner Griswold for your kindness and for making these projects and the podcast possible through your donations.
If you’d like to support these projects like this, check out this page.
If you liked this, follow me on

References
Demb, J. B., & Singer, J. H. (2012). Intrinsic properties and functional circuitry of the AII amacrine cell. Visual neuroscience, 29(1), 51.
Fosser, N. S., Brusco, A., & Ríos, H. (2005). Darkness induced neuroplastic changes in the serotoninergic system of the chick retina. Developmental brain research, 160(2), 211-218.
Gloriani, A. H., & Schütz, A. C. (2019). Humans trust central vision more than peripheral vision even in the dark. Current Biology, 29(7), 1206-1210.
Inserra, A., De Gregorio, D., & Gobbi, G. (2021). Psychedelics in psychiatry: neuroplastic, immunomodulatory, and neurotransmitter mechanisms. Pharmacological Reviews, 73(1), 202-277.
JIN, X. T., & BRUNKEN, W. J. (1998). Serotonin receptors modulate rod signals: a neuropharmacological comparison of light-and dark-adapted retinas. Visual neuroscience, 15(5), 891-902.
Ly, C., Greb, A. C., Cameron, L. P., Wong, J. M., Barragan, E. V., Wilson, P. C., … & Olson, D. E. (2018). Psychedelics promote structural and functional neural plasticity. Cell reports, 23(11), 3170-3182.
Masland, R. H. (2017). Vision: two Speeds in the Retina. Current Biology, 27(8), R303-R305.
Masson, J. (2019). Serotonin in retina. Biochimie, 161, 51-55.
Purves, D., Augustine, G. J., Fitzpatrick, D., Katz, L. C., LaMantia, A. S., McNamara, J. O., & Williams, S. M. (2001). Functional specialization of the rod and cone systems. Neuroscience, 2.
Enjoyed to read. Thanks for share!
LikeLike